Formulating Questions about Household Materials:
Observing everyday materials helps us understand their physical and chemical properties. Asking the right questions encourages scientific curiosity and inquiry-based learning.
Examples of Household Materials
- Fruits and vegetables: Lemon, apple, tomato
- Spices and condiments: Salt, turmeric, sugar
- Liquids: Water, oil, vinegar
- Other items: Cloth, paper, soap
| Importance of Questions | Examples of Questions |
|
Understand properties of materials
|
Why is lemon sour?
|
|
Investigate reasons for observations
|
Does sugar dissolve faster in hot water?
|
|
Develop scientific thinking
|
Why does milk turn sour if left outside?
|
Steps to Formulate Good Questions:
- Observe the material carefully (color, texture, taste)
- Record your observations
- Ask why and how questions
- Make predictions based on prior knowledge
- Plan safe experiments to test your questions
Predicting and Observing Chemical Reactions in Stain Removal:
Household stains, such as turmeric on fabric, involve chemical compounds. Cleaning agents can cause chemical or physical reactions that remove or change the stain.
Example: Turmeric Stain
- Contains the yellow pigment curcumin, which is not easily soluble in water.
- Cleaning agents interact with the pigment through different reactions.
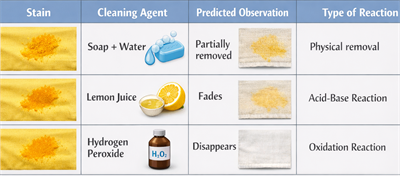
Predictions Table
Observation Steps
- Apply a turmeric stain to fabric.
- Treat with a chosen cleaning agent.
- Observe color change or removal of stain.
- Record the results accurately.
Explanation of Reactions
- Soap/Detergent: Removes pigment physically (emulsification)
- Acid (lemon juice): Reacts with curcumin, causing fading
- Oxidizing agent (H₂O₂): Breaks down pigment permanently
Classifying Materials into Metals and Non-Metals:
Metals
- Properties: Shiny, malleable, ductile, hard, good conductors of heat & electricity, sonorous
- Examples: Iron, Copper, Aluminium, Gold
Non-Metals
- Properties: Dull, brittle, poor conductors, soft or gaseous, non-sonorous
- Examples: Sulfur, Phosphorus, Oxygen, Chlorine
Comparison Table
| Property | Metals | Non-Metals |
| Appearance | Shiny | Dull |
| Malleability | Yes | No |
| Ductility | Yes | No |
| Conductivity | Good | Poor |
| Hardness | Hard | Soft/Brittle |
| Sonority | Sonorous | Non-Sonorous |
Electrical Properties of Materials and Bulb Glow:
A bulb glows in a simple circuit only if the material allows electric current to flow. Observing bulb glow helps classify materials as conductors or insulators.
| Conductors | Insulators |
| Materials that allow electric current to pass easily. | Materials that do not allow electric current to pass. |
| Copper, Aluminium, Iron | Plastic, Rubber, Wood, Glass |
| Bulb glows in a circuit with a conductor | Bulb does not glow in a circuit with an insulator |

An electric circuit
Classifying Materials using Bulb Glow:
| Materials | Bulb Glows? | Classification |
| Copper Wire | Yes | Conductor |
| Aluminium Foil | Yes | Conductor |
| Plastic Rod | No | Insulator |
| Wooden Stick | No | Insulator |