Difference between atoms and molecules:
|
Atom
|
Molecule
|
|
An atom is the smallest particle of an element.
|
A molecule is the smallest particle of an element or compound.
|
| Except in noble gases, atoms do not exist in free state. | Molecule exists in a free state. |
| Except for some noble gases, other atoms are highly reactive. | Molecules are less reactive. |
|
Chemical bonds do not exist between atoms.
|
Atoms in a molecule are held by chemical bonds. Example: Water molecule.
|
Mole:
- In the SI system, the mole (or mol) is the amount of a substance that contains as many elementary entities (atoms, molecules, or other particles) as there are atoms in exactly \(12\ g\) (or \(0.012\ kg\)) of the carbon - \(12\) isotope.
- Experimentally, the number of atoms in \(12\) \(g\) of carbon-\(12\) is determined. This is called 'Avogadro's number' (\(N_A\)), named after an Italian scientist Amedeo Avogadro who proposed its significance. Its value is \(6.023\times10^{23}\).
- Hence, one mole of a substance contains \(6.023\times10^{23}\) entities. Thus, \(5\) moles of oxygen molecules contain \(5\times6.023\times10^{23}\) molecules.
PYQ - Mole
Mole concept:
The study of the collection of particles by using mole as the counting unit, in order to express the mass and volume of such unit particles in bulk of matter is known as mole concept. The number of moles of a substance can be determined in several of ways depending on the data available, as follows:
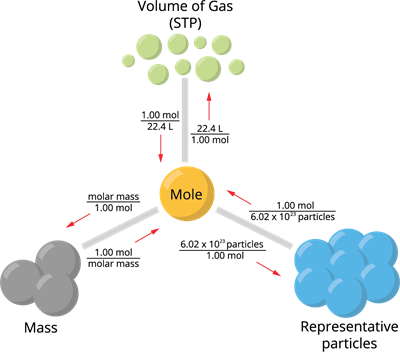
Mole concept
Calculating number of moles from mass and volume:
| 1. | Calculate the number of moles in \(46\) \(g\) of sodium. |
Data:
Given mass of sodium \(= 46\) \(g\).
Atomic mass or mass number of sodium \(= 23\)
The formula to find the number of moles from the mass is,
Hence, the number of moles in \(46\) \(g\) of sodium \(=2\) \(moles\)
| 2. | Calculate the number of moles in \(5.6\) \(L\) of oxygen at STP. |
Given volume of oxygen \(= 5.6\) \(L\)
Molar volume of oxygen at STP \(= 22.4\) \(L\)
The formula to find the number of moles from the volume is,
Hence, the number of moles in \(5.6\) \(L\) of oxygen at STP\(=0.25\) \(moles\)
| 3. | Calculate the number of moles of a sample that contains \(12.046\times10^{23}\) atoms of iron. |
Data:
Given the number of atoms of iron \(=12.046\times10^{23}\)
Avogadro's number \(=6.023\times10^{23}\)
Hence, the number of moles of a sample that contains \(12.046\times10^{23}\) atoms of iron\(=2\) \(moles\)
Calculation of mass from Avogadro number:
| 1. | Calculate the mass of \(1.51\times10^{23}\) molecules of water. |
Data:
Given molecules of \(H_2O=1.51\times10^{23}\)
Molecular mass of \(H_2O=18\)
Avogadro's number \(=6.023\times10^{23}\)
Hence, the mass of \(1.51\times10^{23}\) molecules of water \(=4.5\) \(g\)
| 2. | Calculate the mass of \(5\times10^{23}\) molecules of glucose. |
Data:
Given molecules of glucose \(=1.51\times10^{23}\)
Molecular mass of glucose \(=180\)
Avogadro's number \(=6.023\times10^{23}\)
Hence, the mass of \(5\times10^{23}\) molecules of glucose \(=149.43\) \(g\)
PYQ - Calculation of moles
Avogadro's law:
Avogadro’s law states that “equal volumes of all gases under similar conditions of temperature and pressure contain the equal number of molecules”.
As a result, the volume of any gas must be proportional to the number of molecules present in it. If '\(V\)' is the volume and '\(n\)' is the number of molecules in a gas, then Avogadro law is written as follows:
PYQ - Avogadro’s law
Applications of Avogadro's law:
- It describes the law of Gay-Lussac.
- It helps in identifying the atomicity of gases.
- Avogadro's law can be used to find the molecular formula of gases.
- The relation between molecular mass and vapour density is determined by it.
- It helps to calculate the gram molar volume of all gases (i.e., \(22.4\) litre at STP).
Percent composition:
The percentage composition of a compound represents the mass of each element present in \(100\) \(g\) of the compound.
PYQ - Percent composition
Relationship between vapour density and relative molecular mass:
- Vapour density is the ratio of the mass of a certain volume of a gas or vapour, to the mass of an equal volume of hydrogen, measured under the same conditions of temperature and pressure.
- The relative molecular mass of a gas or vapour is the ratio between the mass of one molecule of the gas or vapour to the mass of one atom of hydrogen.
According to Avogadro's law, all gases have the same number of molecules in equal volumes. Thus, let the number of molecules in one volume \(= n\), then cancelling '\(n\)' which is common and since hydrogen is a diatomic,
When you compare the formula for vapour density and relative molecular mass, and by substituting the values, we get,
or