Nuclear physics is the study of atomic nuclei and their constituents (protons and neutrons).
Atom:
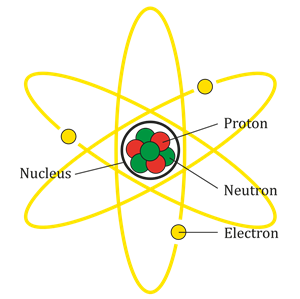
Atoms are mainly composed of particles such as electrons, protons, and neutrons. In an atom, the negatively charged particles are the electrons, the positively charged particles are the protons, and the chargeless particles are the neutrons.
Thus, Henri Becquerel found that he could replicate the same effect if he placed Uranium near a photographic film. It was clear that the Uranium emitted something that may alter a photographic plate. Hence, this phenomenon was termed radioactivity, and Uranium was a radioactive element.
Nuclear decay is a process that converts these unstable nuclei into more stable nuclei.
Radioactivity is the phenomenon of nuclear decay of certain elements with the emission of radiations like alpha, beta, and gamma rays. It is the phenomenon of self-disintegration and spontaneous emission of radiation by some unstable nuclei with atomic numbers greater than 82.
Types of radioactivity:
The two types of radioactivity are
- Natural radioactivity
- Artificial radioactivity
The particle that induces the artificial disintegration is known as the projectile, and the particle produced after the disintegration is known as the ejected particle.
Artificial transmutation is the process of transformation of one element into another element by artificial disintegration means of colliding it with high energy particles.
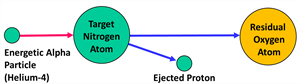
Artificial transmutation by alpha particle
Artificial transmutation of fluorine and aluminium is possible by using the α-particles as projectiles.
Artificial radioactivity, also known as man-made radioactivity, is the phenomenon by which lighter elements are made radioactive by artificial or induced methods.
Some of the units of radioactivity are:
- Curie
- Rutherford
- Becquerel
- Roentgen
Curie:
Curie is the quantity of a radioactive substance that undergoes \(3.7 \times 10^{10}\) disintegrations in one second.
The value is close to the activity of \(1\ g\) of radium \(226\). Curie is the oldest unit of radioactivity.
\(1\ curie\) \(=\) \(3.7 \times 10^{10}\) \(disintegrations\ per\ second\)
Rutherford:
Another unit of radioactivity is Rutherford.
Rutherford (\(Rd\)) is the quantity of a radioactive substance, which produces \(10^{6}\) disintegrations in one second.
Becquerel:
Becquerel (\(Bq\)) is defined as the quantity of one disintegration per second. The SI unit of radioactivity is becquerel.
Roentgen:
One roentgen (\(R\)) is the quantity of radioactive substance which produces a charge of \(2.58 \times 10^{-4}\) \(coulomb\) in \(1\ kg\) of air under standard conditions of pressure, temperature and humidity
- Alpha (α) rays
- Beta (β) rays
- Gamma (γ) rays

Alpha rays are the helium nucleus (\(_{2}He^{4}\)) consisting of two protons and two neutrons. They are positively charged particles, having a charge of \(+2e\).
Beta (\(β\)) rays are the electrons (\(_{-1}e^{0}\)), one of the fundamental elementary particles in an atom. They are negatively charged particles, having a charge of \(–e\).
Gamma (\(γ\)) rays are electromagnetic waves consisting of photons.
Alpha decay:
Beta decay:
Gamma decay:
The law states that ‘in any radioactive substance, the number of nuclei disintegrating per second is directly proportional to the number of nuclei present’.
Half-life period:
The half-life period (\(T\)) of a radioactive substance is defined as the time during which half the amount of the substance disintegrates.
This equation helps to find the mass of un-decayed particles.