Kindly check out the below video to learn about Solutions. Watch the video till the end to complete this task.
Importance:
The chapter "Solutions” is an important topic in chemistry and carries a weightage of \(4\) to \(5\) marks.
It helps students understand the concept of solutions based on,
- Different types of solutions,
- Solubility and factors affecting solubility,
- Concentration of solutions and basic calculations,
- Hydrated and anhydrous salts, and the difference between deliquescent and hygroscopic substances.
Question distribution:
- Part I (\(1\) mark) – One question
-
Part II (\(2\) mark) – One question
-
Part III (\(4\) mark) – One question
(Note: The exact mark distribution may vary slightly across examinations.)
Learning outcomes:
- Concept of solution: Understand the meaning and components of a solution (solute and solvent).
- Types of solutions: Learn the classification of solutions based on the physical states of solute and solvent.
- Solubility: Understand solubility and the factors affecting solubility of solids and gases in liquids.
- Concentration of solutions: Learn different methods of expressing concentration and perform basic calculations.
- Salts and their properties: Understand hydrated and anhydrous salts and distinguish between deliquescent and hygroscopic substances.
Introduction to mixtures:
A mixture is a substance made of two or more elements or compounds or both, physically mixed in any ratio.
Solution and its components:
A solution is a homogeneous mixture of two or more substances that appears to be uniform in appearance..
- A solute is a part of a solution that is present in a smaller quantity by weight.
- A solvent is a variable that is present in a greater quantity by weight.
Binary solution: A solution having one solute and one solvent is called a binary solution. For example, copper sulphate solution
Ternary solution: A solution having three components is called a ternary solution. For example, adding mixture of salt and sugar to water
Types of solutions based on physical state of solute and solvent:
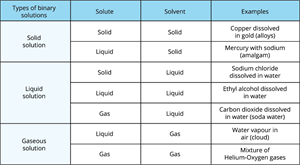
Different types of binary solutions
However, the solvent accounts for the majority of the solution. The physical state is the primary factor that determines the characteristics of the solution.
Types of solutions based on the type of solvent:
- Aqueous solutions: It is a solution in which water acts as the solvent. For example, common salt in water, sugar in water, copper sulphate in water.
- Non-aqueous solutions: It is a solution in which any liquid other than water acts as the solvent. For example, iodine dissolved in carbon tetrachloride, sulphur dissolved in carbon disulphide.
PYQ - Solvent
Types of solutions based on the amount of the solute:
- Saturated solution: A saturated solution is one in which no more solute can be dissolved in a specific amount of solvent at a given temperature.
- Unsaturated solution: At a given temperature, an unsaturated solution contains less solute than a saturated solution.
- Supersaturated solution: At a given temperature, a supersaturated solution contains more solute than a saturated solution.
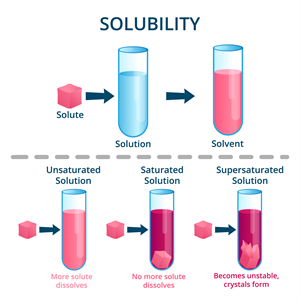
Types of Solutions Based on Solubility
Types of solutions based on the amount of solute in the solution:
- Dilute solution: Dilute solution refers to a solution with a minimal volume of solute. This can also be known as an unsaturated solution.
- Concentrated solution: A concentrated solution is a solution that contains a significant volume of solute.
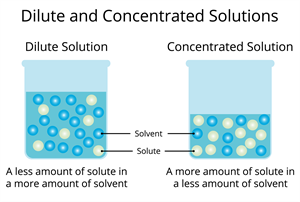
Types of solution based on solute
Solubility:
Solubility is defined as the number of grams of a solute that can be dissolved in \(100g\) of a solvent to form its saturated solution at a given temperature and pressure.
The solubility is expressed mathematically as
PYQ - Solubility
Factors affecting solubility:
-
Nature of solute and solvent:"Like dissolves like.” Polar solutes dissolve in polar solvents (e.g., salt in water), and non-polar solutes dissolve in non-polar solvents. For example, fat dissolved in ether.
-
Temperature:
-
Solids in liquids: Solubility usually increases with temperature. For example sugar in warm water.
-
Gases in liquids: Solubility decreases with temperature, so gases escape as bubbles when water is heated. For example, aquatic animals thrive in cold regions because cold water holds more dissolved oxygen than warm water.
-
-
Pressure:
Pressure affects mainly gases in liquids. Higher pressure increases gas solubility. For example, carbonated beverages.
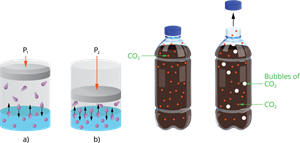
Effect of pressure on solubility
Concentration of a solution:
The concentration of a solution may be defined as the amount of solute present in a given amount of solution or solvent.
There are various ways to express the concentration of a solution, but here, we will learn these two well-known methods:
1. Mass percentage: It is defined as the percentage by mass of the solute present in the solution. It is typically used when the solute is solid, and the solvent is liquid.
PYQ - Mass percentage
2. Volume percentage: It is defined as the percentage of solute (in \(mL\)) present in a given volume of solution.
Hydrated salts and water of crystallisation:
The number of water molecules found in the crystalline substance is called water of crystallisation. These salts are known as hydrated salts.
Common hydrated salts:
|
Common Name
|
IUPAC Name
|
Molecular formula
|
|
Blue Vitriol
|
Copper (II) sulphate pentahydrate
|
\(CuSO_4.5H_2O\)
|
|
Epsom Salt
|
Magnesium sulphate heptahydrate
|
\(MgSO_4.7H_2O\)
|
|
Gypsum
|
Calcium sulphate dihydrate
|
\(CaSO_4.2H_2O\)
|
|
Green Vitriol
|
Iron (II) sulphate heptahydrate
|
\(FeSO_4.7H_2O\)
|
|
White Vitriol
|
Zinc sulphate heptahydrate
|
\(ZnSO_4.7H_2O\)
|
Reaction of Hydrated Copper Sulphate on Heating:
PYQ - Water of crystallisation
Hygroscopy and deliquescence:
|
Hygroscopic substance
|
Deliquescence substance
|
| They absorb moisture but do not dissolve when exposed to the atmospheric air at room temperature. | They absorb moisture and dissolve when exposed to ordinary atmospheric air at room temperature. |
| When exposed to air, hygroscopic substances do not change their physical state. | When deliquescent substances are exposed to air, their physical state changes. |
| They may be amorphous solids or liquids. | They are crystalline solids. |
PYQ - Hygroscopic and deliquescence