Temperature:
The measurement of the hotness or coldness of a substance is known as its temperature. It determines the direction of the flow of heat when two bodies are placed in contact.
Units of Temperature:
Three units are used to measure the temperature:
-
Degree Celsius
-
Fahrenheit and
-
Kelvin
Fahrenheit: Fahrenheit is written as °F.
Kelvin: Kelvin is written as K.
Measuring Temperature:
The temperature of the substance is the approximation of the average kinetic energy of the substance. Higher temperature means the molecules inside the substance are moving rapidly; similarly, lower temperature means that the molecules are moving in a slow phase.
Solids, Liquids and Gases are affected by heat. It expands when heated and contracts when cooled. This principle is used to measure the temperature. The thermometer is used to measure the temperature.
Temperature Scales:
|
Temperature
|
Kelvin Scale (K)
|
Celsius Scale (ºC)
|
Fahrenheit Scale (ºF)
|
| Absolute Zero |
0
|
-273.15
|
-459.67
|
| Boiling point of water |
373.15
|
100
|
212
|
| Freezing point of water |
273.15
|
0
|
32
|
| Mean temperature of human body |
310.15
|
37
|
98.6
|
| Room temperature (average) |
296.15
|
23
|
72
|
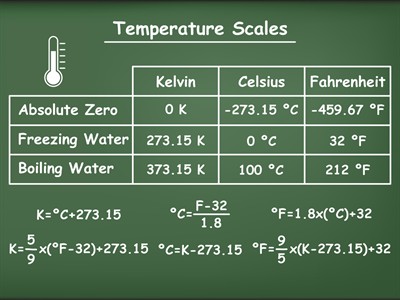
Relationships between the temperature scales
Heat:
Heat is a form of energy that flows from a body at higher temperature to a body at lower temperature. It is transferred due to a temperature difference between two objects.
Heat depends on both the temperature of the substance and the number of particles present in it. It is related to the total kinetic energy of all the molecules in a substance.
Heat is measured in joules (J) in the SI system and also in calories. A calorimeter is used to measure heat, and heat has the ability to do work.
Heat is measured in joules (J) in the SI system and also in calories. A calorimeter is used to measure heat, and heat has the ability to do work.
Temperature:
Temperature is the measure of the degree of hotness or coldness of a body.
It determines the direction of heat flow, as heat always flows from higher to lower temperature.
It determines the direction of heat flow, as heat always flows from higher to lower temperature.
Temperature depends on the average kinetic energy of the molecules of a substance.
It does not depend on the number of particles present in the object.
The SI unit of temperature is kelvin (K), and it is also measured in Celsius (°C) and Fahrenheit (°F). Temperature is measured using a thermometer.
Thermal equilibrium:
Whenever two systems come into thermal contact, one system's energy is transferred to another to reach thermal equilibrium. Here, heat or thermal energy is transferred between the objects. Generally, the temperature determines the direction of flow. In this case, the heat is transferred from the hotter object to the colder object.
Heat:
Heat is a scalar quantity. The SI unit of heat energy absorbed or evolved is the joule (J).
Other units of Heat energy:
Calorie:
One calorie is defined as the amount of heat or thermal energy required to increase the temperature of \(1\ gram\) of water through \(1\ °C\).
Kilocalorie:
One kilocalorie is defined as the amount of heat or thermal energy required to increase the temperature of \(1\ kilogram\) of water through \(1\ °C\).
This process of heat transfer may be done in any of the following ways,
- Conduction
- Convection and
- Radiation
Effects of Heat energy:
Whenever the heat energy is supplied to any substance, it increases its temperature and also brings more changes.
The following are the three important effects of heat that we can see in our daily life,
- Thermal expansion in objects
- Increase in temperature of the object
- Change in state of the substance
Changes of Phase:
The term change of phase means the same thing as the term change of state. The change of phase always occurs with a change of heat. If heat energy is supplied to or taken out from a substance, it will undergo a change from one state of matter to another state. However, the temperature does not change.
|
Description of Phase change
|
Term for Phase change
|
Heat movement during Phase change
|
Temperature change during Phase change
|
|
Solid to Liquid
|
Melting
|
Heat goes into the solid as it melts.
|
None
|
|
Liquid to Gas
|
Vaporization, which includes boiling and evaporation
|
Heat goes into the liquid as it vaporizes.
|
None
|
|
Solid to Gas
|
Sublimation
|
Heat goes into the solid as it sublimates.
|
None
|
Thermal Expansion:
The expansion of a substance on heating is called, the thermal expansion of that substance. The change in length, area or volume due to contraction or expansion is directly proportional to temperature change.
Types of expansion in solids:
Whenever we heat a solid, there is an increase in the dimensions of the body. It is known as expansion in solids.
It undergoes three types of expansions.
Linear or Longitudinal expansions:
When we heat a solid, if there is an increase in the body's length, this is called Linear expansion. It is also known as Longitudinal expansion.
Areal or Superficial expansions:
When we heat a solid, if there is an increase in the area of the body, this is called Areal expansion. It is also known as Superficial expansion.
Cubical or Volumetric expansions:
When we heat a solid, if there is an increase in the volume of the body, this is called Cubical expansion. It is also known as Volumetric expansion.
Expansion in liquid:
When a liquid is heated, the energy of the atoms in a liquid or gas increases, and they are forced further apart. The amount of expansion in liquids and gases vary from substance to substance depending on their properties.
The coefficient of cubical expansion of liquid does not depend on its temperature, whereas its value for gases mainly depends on the temperature of gases.
Hence, for liquids, we can define,
- Real expansion and
- Apparent expansion
Real expansion:
The coefficient of real expansion is defined as the ratio of the actual rise in the liquid's volume per degree rise in temperature to its unit volume.
The SI unit of coefficient of real expansion is \(K^{−1}\).
Apparent expansion:
The coefficient of apparent expansion is defined as the ratio of the apparent rise in the liquid's volume per degree increase in temperature to its unit volume.
The SI unit of coefficient of apparent expansion is \(K^{−1}\).
Expansion in gas:
Gas is one of the states of matter that has no fixed shape and volume. Gases have a lower density compared to other states of matter, such as solids and liquids.
There is a wide range of space between particles, with a higher kinetic energy than the attractive forces between them. The particles move very fast and interact with others, causing them to diffuse or spread out until they are uniformly distributed throughout the volume of the container.
The gas laws consist of three primary laws:
- Charles' law
- Boyle's law and
- Avogadro's law
Gases are categorised into,
- Real gases and
- Ideal gases
Real Gases:
A real gas behaves like an ideal gas at a very high temperature or low pressure because there is no interatomic or intermolecular force of attraction between molecules or atoms in this condition.
Ideal Gases:
If the atoms or molecules of a gas do not interact or collide, then the gas is called an ideal gas or a perfect gas.
Ideal gases obey Boyle’s law, Charles’s law and Avogadro’s law. All these laws state the relationship between various properties of a gas such as pressure (P), volume (V), temperature (T) and the number of atoms (n).
Boyle's Law:
When the temperature of a gas is kept constant, the volume of a fixed mass of gas is inversely proportional to its pressure.
Boyle’s Law can be used to determine the current pressure or volume of gas and can be represented also as
Charles's Law:
The volume (V) of a given mass of a gas, at constant pressure (P), is directly proportional to its absolute temperature (T).
Avogadro's law:
It states that the volume occupied by an ideal gas is directly proportional to the number of atoms or molecules of the gas present in the container at constant pressure and temperature.
Ideal gas equation:
The ideal gas equation is an equation, which relates all the properties of an ideal gas.
An ideal gas obeys Boyle’s law and Charles’ law, and Avogadro’s law.
According to Boyle’s law,
----- (Eq. 1)
According to Charles’s law,
----- (Eq. 2)
According to Avogadro’s law,
----- (Eq. 3)
Combining equations (Eq. 1), (Eq. 2) and (Eq. 3),
The above equation shows that the volume of a gas (\(V\)) is proportional to the number of moles (\(n\)) and the temperature (\(T\)) and is inversely proportional to the pressure (\(P\)). This expression can also be written as,
Rearranging the equation,
----- (Eq. 4)
The above relation is called the combined law of gases.
Consider a gas, which contains moles of the gas. The number of atoms contained will be equal to times the Avogadro constant, . The value of Avogadro numbers is .
That is,
----- (Eq. 5)
Substitute Eq. 5 to Eq. 4. And, then the Eq. 4 can be written as,
----- (Eq. 6)
The value of the constant in the above equation is taken to be , which is called as Boltzmann constant ().
Hence, we have the following equation:
----- (Eq. 7)
Rearranging the above equation,
----- (Eq. 8)
Here, , which is termed as universal gas constant whose value is .
Substitute the value of \(R\) in Eq. 8,
The ideal gas equation is also called the equation of state because it gives the relation between the state variables and is used to describe the state of any gas.