Importance:
The chapter "Types of chemical reactions” is an important topic in chemistry and carries a weightage of about \(5\) to \(7\) marks.
It helps students understand the fundamental ideas related to chemical changes, such as:
-
Meaning and representation of chemical reactions
-
Different types of chemical reactions
-
Rate of chemical reactions and factors affecting it
-
Concept of chemical equilibrium
-
pH scale and its role in everyday life
Question distribution:
- Part I (\(1\) mark) - One question
-
Part II (\(2\) mark) - One question
-
Part III (\(4\) mark) - One question
- Part IV (\(7\) mark) - One question
(Note: The exact mark distribution may vary slightly across examinations.)
Learning outcomes:
-
Chemical reactions: Understand the concept of chemical reactions and how they are represented using balanced chemical equations.
-
Types of chemical reactions: Identify and classify chemical reactions into combination, decomposition, displacement, double displacement, combustion reactions, reversible and irreversible reactions.
-
Rate of reaction: Define the rate of a chemical reaction and explain the factors affecting it such as concentration, temperature, catalyst, pressure, and surface area.
-
Chemical equilibrium: Explain the equilibrium state of a reversible reaction and list the characteristics of chemical and physical equilibrium.
-
pH and ionic product of water: Define pH and ionic product of water and relate pH values to acidic, basic, and neutral solutions.
-
Role of pH in everyday life: Recognize the importance of pH in digestion, tooth decay, soil pH, acid rain, and the survival of plants and animals.
Chemical reaction
A chemical reaction is a process in which the atoms of reactant molecules are rearranged by breaking old chemical bonds and forming new bonds to produce new substances. During this process, energy is absorbed during bond breaking and released during bond formation. Whether a reaction is exothermic or endothermic depends on the overall energy change.
Types of chemical reactions
Combination Reaction:
A combination reaction is a reaction in which two or more substances combine to form a single product. Most combination reactions are exothermic and is classified into three types based on the chemical nature of the reactants.
i. \(Element\) + \(Element\) → \(Compound\)
Example: \(2Na\) + \(Cl_2\) → \(2NaCl\)
ii. \(Compound\) + \(Element\) → \(Compound\)
Example: \(PCl_3\) + \(Cl_2\) → \(PCl_5\)
iii. \(Compound\) + \(Compound\) → \(Compound\)
Example: \(SiO_2\) + \(CaO\) → \(CaSiO_3\)
PYQ - Combination reaction
Decomposition reaction:
A decomposition reaction is one in which a single compound splits into two or more simpler substances using energy, and it is mostly endothermic. Based on the energy source, it is classified into three types:
i. Thermal decomposition: By heat
Example: \(CaCO_3\) → \(CaO\) + \(CO_2\)
ii. Electrolytic decomposition: By electricity
Example: \(2NaCl\ {(molten)}\) → \(2Na\) + \(Cl_2\)
iii. Photo decomposition: By light
Example: \(2AgBr\) → \(2Ag\) + \(Br_2\)
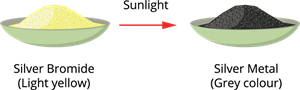
Photodecomposition of silver bromide
Single Displacement Reaction:
A single displacement reaction is a reaction in which a more reactive element displaces a less reactive element from its compound in solution or molten state.
Example: \(Zn\) + \(2HCl\) → \(ZnCl_2\) + \(H_2\)
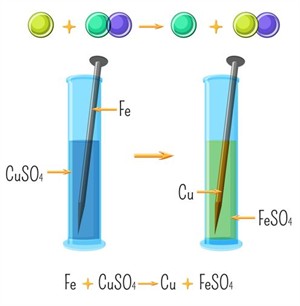
Single displacement reaction
Double displacement reaction:
The exchange of ions between the reactants to form new products. Double displacement reaction has the following two types:
i. Neutralisation reaction:

Neutralisation reaction
PYQ - Neutralization reaction
ii. Precipitation reaction:

Example: \(Pb(NO_3)_2{(aq)}\) + \(2KI{(aq)}\) → \(PbI_2{(s)}\) + \(2KNO_3{(aq)}\)
Reactants: Both are colourless solutions
Products: Yellow precipitate \(PbI_2\)
Combustion reaction:
A combustion reaction is a chemical reaction in which a substance reacts rapidly with oxygen to produce heat and light. The nature of the products formed depends on the substance undergoing combustion. Since heat is released, combustion reactions are exothermic reactions.
Example: \(C_3H_8\) + \(5O_2\) → \(3CO_2\) + \(4H_2O\) + \(Heat\)

Examples of combustion reaction
Classification based on the direction of the reaction:
|
Reversible reaction
|
Irreversible reaction
|
|
Under suitable conditions, it can be reversed.
|
It cannot be reversed.
|
|
Both forward and backward reactions occur
simultaneously. |
It is unidirectional. This reaction occurs only in the forward direction.
|
|
It attains equilibrium.
|
It does not attain equilibrium.
|
|
It is not possible to convert reactants completely into products.
|
It is possible to convert reactants completely into products.
|
|
It is a slow process.
|
It is fast.
|
| This process results in a temporary change. | This process results in a permanent change. |
| Example: \(PCl_5{(g)}\) ⇄ \(PCl_3{(g)}\) + \(Cl_2{(g)}\) | Example: \(C{(s)}\) + \(O_2{(g)}\) → \(CO_2{(g)}\) + \(Heat\) |
Rate of a chemical reaction:
Rate of a reaction is the change in the amount or concentration of any one of the reactants or products per unit time.
Factors influencing the rate of a chemical reaction:
i. Nature of the reactants
ii. Concentration of the reactants
iii. Temperature
iv. Catalyst
v. Pressure (only in reactions involving gases)
vi. Surface area of the reactants
ii. Concentration of the reactants
iii. Temperature
iv. Catalyst
v. Pressure (only in reactions involving gases)
vi. Surface area of the reactants
State of equilibrium:
At equilibrium, products and reactants are formed at equal rates, so their concentrations remain constant.
At equilibrium: \(Rate\) \(of\) \(forward\) \(reaction\) = \(Rate\) \(of\) \(backward\) \(reaction\).
Characteristics of Equilibrium:
- Forward and backward reaction rates are equal in chemical equilibrium.
- The system's observable properties, such as pressure, concentration, colour, density, and viscosity, remain unaffected over time.
- The chemical equilibrium is a dynamic equilibrium since both the forward and backward reactions continue to occur even though it appears static externally. The volume of all phases is constant in physical equilibrium.
Ionic product of water:
The product of the concentration of the hydronium ion and the hydroxyl ion is called ionic product of water. It is denoted by '\(K_w\)'. Mathematically it is expressed as follow:
\(K_w\) = \([H^+]\) \([OH^−]\)
Its unit is \(mol^2\) \(dm^{−6}\). At \(25°C\), its value is \(1.00\) × \(10\) − \(14\).
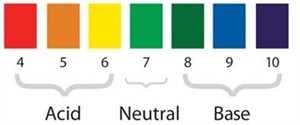
pH scale:
The \(pH\) scale is a set of numbers that ranges from \(0\) to \(14\) and is used to determine whether a solution is acidic, basic, or neutral.
| Type of solution | pH range |
|
Acids
|
\(< 7\)
|
|
Neutral solutions
|
\(7\)
|
|
Base
|
\(> 7\)
|

Universal indicator \(pH\) chart
Role of pH in everyday life:
-
Blood: The pH of blood ranges from \(7.35\) to \(7.45\), and even a small change can lead to diseases.
-
Digestive system: The pH of stomach acid is about \(2.0\), which helps in digestion, but excess acid causes indigestion.
-
Tooth decay: When the \(pH\) of saliva falls below \(5.5\), tooth enamel gets damaged, basic toothpaste helps neutralise the acid.
-
Soil pH: Different crops require different soil \(pH\). Citrus grows in slightly alkaline soil, rice in acidic soil, and sugarcane in neutral soil.
-
Rainwater: Pure rainwater has a \(pH\) of about \(5.6\) due to dissolved carbon dioxide, but acid rain (\(pH\) less than \(5.6\)) harms aquatic life.pH and pOH calculations:The \(pH\) is the negative logarithm of the hydrogen ion concentration.i.e, \(pH\) = \(−log_{10}\) \([H^+]\)Example: Calculate the \(pH\) of \(0.01\) \(M\) \(HNO_3\).
\([H^+]\) = \(0.01\)
\(pH\) = \(−log_{10}\) \([H^+]\)
\(pH\) = \(−log_{10} [0.01]\)
\(pH\) = \(−log_{10} [1 × 10^{−2}]\)
\(pH\) = \(−(log_{10}1 − 2 log_{10}10)\)
\(pH\) = \(0 + 2 × log_{10}10\)
\(pH\) = \(0 + 2 × 1 = 2\)
\(pH\) = \(2\)PYQ - pH calculationThe \(pOH\) is the negative logarithm of the hydroxyl ion concentration.\(pOH\) = \(−log_{10}[OH^−]\)Example: The hydroxyl ion concentration of a solution is \(1 × 10^{−9} M\). Calculate the \(pOH\) of the solution.\(pOH\) = \(−log_{10}[OH^−]\)\(pOH\) = \(−log_{10}[1 × 10^{−9}]\)
\(pOH\) = \(−(log_{10} × 1.0 + log_{10} × 10^{−9})\)
\(pOH\) = \(−(0 − 9log_{10}10)\)
\(pOH\) = \(−(0 − 9)\)
\(pOH\) = \(9\)Relationship between \(pH\) and \(pOH\):At \(25°C\), \(pH\) + \(pOH\) = \(14\)Example: \(pOH\) of a solution is \(11.76\). What is the \(pH\) of this solution?\(pH\) = \(14\) − \(pOH\)
\(pH\) = \(14 – 11.76\) = \(2.24\)PYQ - pH value