In this theory, we will learn about the properties of acids and bases, including:
-
Ionisation in water (production of hydrogen and hydroxide ions)
-
Electrical conductivity as electrolytes
-
Effect of water, strength, concentration, and pH
These concepts help us understand how acids and bases behave in aqueous solutions.
Common properties of acids and bases:
-
Acids and bases ionise in aqueous solution, where acids produce \(H^+\) or \(H_3O^+\) ions, while bases produce \(OH^-\) ions.
- Acids and bases show their properties only in the presence of water.
-
Acids and bases conduct electricity in aqueous solution due to the presence of mobile ions and hence act as electrolytes.
-
Acids and bases undergo neutralisation reaction to form salt and water.
Activity: To show that acids and bases conduct electricity
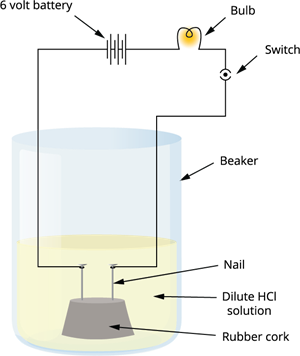
Procedure:
-
Take a solution of dilute hydrochloric acid (\(HCl\)) in a beaker.
-
Fix two iron nails on a cork and place it in the beaker.
-
Connect the nails to a \(6\) \(V\) battery, a bulb, and a switch as shown in the circuit.
-
Switch on the current and observe the bulb.
-
Repeat the experiment using other solutions like glucose, alcohol, sulphuric acid, and sodium hydroxide (\(NaOH\)).

Acid conducts electricity
Observation:
-
The bulb glows in hydrochloric acid and sulphuric acid solutions.
-
The bulb also glows in basic solutions like sodium hydroxide (\(NaOH\)).
-
The bulb does not glow in glucose and alcohol solutions because they do not ionise in aqueous solution.
Result:
Acids and bases conduct electricity in aqueous solution due to the presence of mobile ions, whereas glucose and alcohol do not as they do not ionise.
Acid and base in a water solution:
Acids produce \(H_3O^+\) ions in aqueous solution, which are responsible for conduction. Let us now test to see if these ions are produced only in the presence of an aqueous solution or not with the help of an activity.
Activity: To show that acids produce ions in aqueous solution
Procedure:
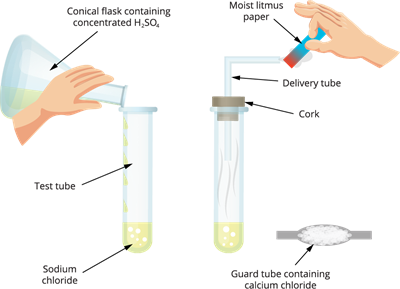
Acid produces \(H^+\) ions in water
Observation:
Procedure:
-
Take about \(1\) \(g\) of solid sodium chloride (\(NaCl\)) in a clean and dry test tube.
-
Add concentrated sulphuric acid (\(H_2SO_4\)) and close the test tube with a rubber cork.
-
Attach a delivery tube to the cork.
-
Observe the gas coming out through the delivery tube.
-
Test the gas using dry blue litmus paper and then wet blue litmus paper.

Acid produces \(H^+\) ions in water
Observation:
-
Dry blue litmus paper shows no change.
-
Wet blue litmus paper turns red.
-
Dry HCl gas: Does not contain free \(H^+\) ions, hence does not show acidic character.
-
HCl solution: Produces \(H^+\) or \(H_3O^+\) ions in water and shows acidic character.
-
Therefore, acids produce \(H^+\) or \(H_3O^+\) ions only in the presence of water.
\(HCl + H_2O → H_3O^+ + Cl^-\) -
Bases also behave similarly by producing \(OH^-\) ions in aqueous solution.
\(NaOH_{(s)}\overset{H_2O}{\rightarrow} Na^+_{(aq)} + OH^-_{(aq)}\)
-
Hence, acids and bases show their properties only in aqueous solutions.
Effect of adding water to acids and bases:
- Dissolving an acid or a base in water is an exothermic process, in which heat is liberated.
- Hence, acid should always be added to water, and not water to acid, to avoid splashing or overheating.
- Adding water to an acid or a base decreases the concentration of hydrogen or hydroxide ions per unit volume.
How strong is acid and base solution:
In order to understand how strong is an acid or a base first, let us see the classification of acids and bases based on the following characteristics
- Strength
- Concentration
1. Strength:
- Strong acid/base: When complete ionisation of acid/ base takes place in an aqueous solution, it is known as strong acid/base. Here the high concentration of \(H^+\) / \(OH^-\) ions is present.
- Weak acid/base: When partial ionisation of acid/base takes place in an aqueous solution, it is known as weak acid/base. Here the low concentration of \(H^+\) / \(OH^-\) ions is present.
2. Concentration:
- Concentrated acid/base: If the aqueous solution contains a high percentage of acid/base and a low percentage of water, it is said to be concentrated acid/base. A concentrated acid/base contains a larger amount of acid/base and hence more \(H^+\) / \(OH^-\) ions per unit volume.
- Diluted acid/base: If the aqueous solution contains a high percentage of water and a low percentage of acid/base, it is said to be dilute acid/base. A dilute acid/base contains a smaller amount of acid/base and fewer \(H^+\) / \(OH^-\) ions per unit volume due to dilution.
pH scale:
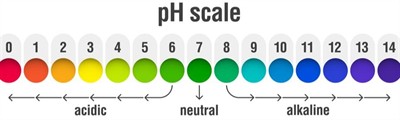
pH scale
-
Acid-base indicators can distinguish acids from bases, but we can also measure the amount of \(H^+\) or \(OH^-\) ions quantitatively.
-
A universal indicator, which is a mixture of several indicators, shows different colours at different concentrations of hydrogen ions.
-
The pH scale measures the hydrogen ion concentration in a solution.
-
“\(p\)” stands for potenz (German for “power”), and "\(H\)" stands for hydrogen.
-
The scale ranges from \(0\) (very acidic) to \(14\) (very alkaline).
-
-
Neutral solution: pH \(=\) \(7\).
-
Acidic solution: pH \(<\) \(7\) (higher \(H^+\) ion concentration decreases the pH).
-
Basic/alkaline solution: pH \(>\) \(7\) (higher \(OH^-\) ion concentration increases the pH).
-
Universal indicator paper is commonly used to determine the \(pH\) of a solution and thus judge its acidic or basic strength.
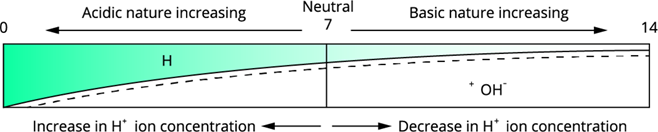
Variation of pH