Everyday life is full of invisible chemical reactions happening around us – from the sour taste of lemon juice to the slippery feel of soap. These reactions are often caused by two special kinds of substances called acids and bases.

Sour acids and soapy base
Get ready to mix solutions, test, and explore the science behind the reactions, indicators, and pH value mysteries — where each solution has a story!
Acids:
The properties of acids are as follows:
-
Acids are sour and corrosive.
-
The pH level of an acid is less than 7.
-
Acids release \(H^+\) ions, which are responsible for their acidic properties.

Properties of acid
Acids are commonly found in various fruits, including:
- Lemon, oranges contain citric acid.
- Apple contains malic acid.
- Grapes have tartaric acid.
The acids that are present naturally are called organic acids, and acids which are synthesised in laboratories, such as \(HCl\), \(HNO_3\) and \(H_2SO_4\), are mineral acids. Generally, organic acids are weak acids, whereas mineral acids are strong acids.
Bases:
The properties of bases are as follows:
- Bases are bitter, and they feel soapy or slippery.
- The pH level of a base is greater than 7.
- Bases that can dissolve in water are known as alkalis, for example, potassium hydroxide and sodium hydroxide.
- Bases release \(OH^-\) ions, which are responsible for their basic properties.

Properties of base
Bases commonly found in cleaning agents include:
- Soap, cleansing products contain sodium hydroxide and potassium hydroxide.
- Baking soda contains sodium bicarbonate.
Acid - base indicators:
The chemical substance that changes colour depending on the pH of the solution, indicating whether the solution is acidic, basic or neutral, is called an acid-base indicator.
i. Litmus paper:
Litmus paper is made from lichens, which are tiny plant-like organisms made of two separate organisms, a fungus and an alga.
Colour change:
- Acidic solution - blue litmus to red
- Basic solution - red litmus to blue
- Neutral solution - both red and blue litmus remain unchanged

Litmus test in different solutions
ii. Phenolphthalein:
Phenolphthalein is a synthetic chemical indicator used to identify acids and bases in a solution.
Colour change:
- Acidic solution - colourless
- Basic solution - pink
iii. Methyl orange:
Methyl orange is a synthetic chemical indicator commonly used to detect acids and bases.
Colour change:
- Acidic solution - red
- Basic solution - yellow
iv. Olfactory indicator:
The olfactory indicators are the substances which indicate a change in odour in acidic and basic media.
Example: Onion, clove oil, garlic extract, and vanilla essence
When these substances are brought into contact with acids and bases, they convey the nature of the chemical, either becoming odourless or retaining the odour.
|
Smell in acid
|
|---|
|
Retains odour
|
|
Retains odour
|
|
Retains odour
|
Reaction of acids with metals:
When an acid reacts with an active metal, it usually produces a salt and hydrogen gas.
\(Metal + Acid → Salt + Hydrogen\ gas\)
Activity: The experiment aims to see how do acids react with metals.
- Step 1: Take a test tube and add \(5\ mL\) of dilute sulphuric acid \(H_2SO_4\) and add a few pinches of zinc granules.
- Step 2: Observe the test tube for any changes. Fit the test tube mouth with a cork and attach one end of the delivery tube to it.
- Step 3: The other end of the delivery tube should be placed inside the glass tube filled with a soap solution.
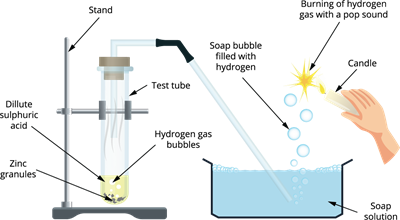
Reaction of dilute sulphuric acid with zinc metal
Observation: The liberation of gas from the reaction passes through the soap solution tub forming bubbles. Since, the hydrogen gas does not dissolve in a soap solution, it escapes.
Confirmation test for the gas released: When a burning candle or splinter is brought near a bubble, the gas burns with a pop sound and extinguishes the flame, which confirms the release of hydrogen gas.
The reaction of zinc with dilute sulphuric acid produces salt and hydrogen gas.
\(Zn + H_2SO_4 \rightarrow ZnSO_4 + H_2\)
Similarly, the reaction of other metals with acids are as follows,
\(Zn + 2HCl \rightarrow ZnCl_2 + H_2\)
\(Fe + 2HCl \rightarrow FeCl_2 + H_2\)
\(Mg + 2HCl \rightarrow MgCl_2 + H_2\)
Not all metals react with acids. Metals that are less reactive than hydrogen such as copper, gold, and silver do not react with dilute acids because they cannot displace hydrogen.
Reaction of bases with metals:
When a reacts with an certain metals (such as zinc or aluminium), it produces a salt and hydrogen gas.
\(Base + Metal → Salt + Hydrogen\ gas\)
Bases (alkalis) such as sodium hydroxide (\(NaOH\)) or potassium hydroxide (\(KOH\)) can react with certain metals to produce salt and hydrogen gas.
When zinc granules are added to sodium hydroxide solution, bubbles of hydrogen gas appear with the formation of complex salt sodium zincate.
\(2NaOH + Zn \rightarrow Na_2ZnO_2+ H_2\)
Sodium hydroxide + Zinc → Salt + Hydrogen gas
Most of the metals do not react with bases. Only amphoteric metals such as zinc, aluminum, and lead, react with bases.