In previous session, we learned about chemical reactions, balancing chemical equations, and the law of conservation of mass. We understood that during a chemical reaction, atoms are neither created nor destroyed; instead, bonds between atoms break and new bonds are formed to produce new substances.
Classification of chemical reactions:
Chemical reactions are classified based on the nature of reactants and products. The major types are:
-
Combination reactions
-
Decomposition reactions
-
Displacement reactions
-
Double displacement reactions
-
Oxidation and reduction reactions
In this session, we will focus on the first two types.
1. Combination reactions:
A combination reaction occurs when two or more reactants combine to form one product. It is also referred to as a synthesis reaction .

Combination reaction
Examples of combination reaction:
i. Formation of slaked lime:
When calcium oxide (quick lime) reacts with water, calcium hydroxide (slaked lime) is formed with the release of heat (exothermic).
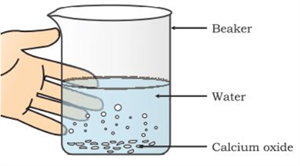
Formation of calcium hydroxide
Observation:
-
A hissing sound is produced
-
The beaker becomes hot
-
Heat is released (exothermic reaction)
\(CaO(s) + H_2O(l) → Ca(OH)_2(aq)\)
Application: Whitewashing of walls
The slaked lime formed reacts slowly with carbon dioxide present in air:
\(Ca(OH)_2 + CO_2 → CaCO_3 + H_2O\)
The formation of calcium carbonate gives walls a shiny and durable finish. Interestingly, marble has the same chemical composition
ii. Burning of coal:
\(C(s) + O_2(g) → CO_2(g)\)
Combustion reactions as examples of combination reactions.
iii. Formation of water:
\(2H_2(g) + O_2(g) → 2H_2O(l)\)
These are also combustion reactions, which release heat and light.
Decomposition reaction:
A single reactant breaks down into two or more products under suitable conditions (current, heat and light).
Note: Most decomposition reactions are endothermic as they require energy to break down compounds. However, the decomposition of organic or vegetable waste into compost is an exothermic process, as it releases heat.

Decomposition reaction
Types of decomposition reaction:
1. Thermal decomposition (by heat):
Example 1: Decomposition of lead nitrate
- \(Pb(NO)_3\): White or colourless solid
- \(PbO\): Yellow residue
- \(NO_2\): Brown fumes
Example 2: Decomposition of calcium carbonate
\(CaCO_3 → CaO + CO_2\)
This reaction is widely used in the cement industry.
2. Electrolytic decomposition (by electricity):
- Gas liberated at the cathode (hydrogen) and the anode (oxygen)
- Confirmation test: Hydrogen (pop sound) and oxygen (glows brightly)
- Volume ratio: \(H_2 : O_2\) \(=2:1\)
- A few drops of \(H_2SO_4\) is added to increase conductivity
3. Photolytic decomposition (by light):
Since the decomposition is caused by light, this reaction is also called photolysis.
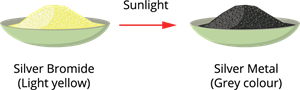
Photolytic decomposition of silver bromide
- Pale yellow silver bromide \(AgBr\) decomposes under light to greyish white silver (\(Ag\)) and bromine (\(Br_2\)).
- Application: Used in black and white photography
Chemical reactions play a vital role in nature and technology. Understanding these reactions helps explain everyday processes like respiration, combustion, and photography.
In the next session, we will explore displacement, double displacement, and redox reactions.