Effect of oxidations reaction in real life:
1. Corrosion:
Corrosion is the progressive destruction of metals by the action of air, moisture or chemicals (such as an acids) on their surface.

Corroded vehicle
Examples:
- When iron is exposed to air and moisture for a long time, it forms a reddish-brown coating called rust (\(Fe_2O_3.xH_2O\)).
\(4Fe + 3O_2 + xH_2O → 2Fe_2O_3 xH_2O\)
- Silver, when exposed to air for a long time, reacts with sulphur compounds present in the air and forms a black coating called tarnish (\(Ag_2S\)).
\(4Ag + O_2 + 2H_2S → 2Ag_2S + 2H_2O\)
- Copper, when exposed to air and moisture for a long time, reacts with carbon dioxide and forms a green coating called patina (basic copper carbonate).
\(2Cu + H_ 2 O + CO_ 2 + O_2 → Cu(OH)_ 2 + CuCO_ 3\)
The green layer formed is basic copper carbonate: \(Cu(OH)_2 + CuCO_3\)
Prevention of corrosion:
- Painting, oiling, greasing, chrome plating, and anodising are all effective methods for preventing metals from rusting.
- Galvanisation is a process that coats steel and iron with a thin layer of zinc to prevent rusting. Even if the zinc coating on the galvanised item is damaged, it remains protected against rust.
- Alloying is an excellent way to improve a metal's properties. This method can be used to obtain the desired properties. When iron is combined with nickel and chromium, stainless steel is formed, which is hard and rust-resistant.
- Electroplating: Coating metals with corrosion-resistant metals like chromium or gold.
2. Rancidity:
The oxidation of oils and fats present in food materials resulting in bad smell and taste is called rancidity.

Rancid bread
When unsaturated (oil/fat) components of food materials are exposed to sunlight, they can be changed into hydroperoxides, which split down into esters, volatile aldehydes, ketones, alcohols, and hydrocarbons (some of which have unpleasant odours).
Oil becomes rancid (rancid oil) due to the decomposition of fats, or sometimes milk becomes rancid due to not heating it in the humid atmosphere.
Methods to prevent rancidity:
- Antioxidants (BHT, BHA, vitamin E, and vitamin C) are typically added to foods containing fats and oils to prevent oxidation.
- We can slow food oxidation by storing it in airtight containers.
- The chips manufacturers usually flush bags of chips with gas like nitrogen to prevent the chips from getting oxidized
Change in temperature:
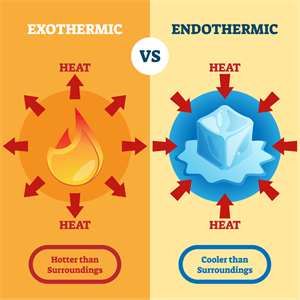
Exothermic vs Endothermic
- Exothermic - Release of heat
- Endothermic - Absorption of heat
- Endothermic examples: Photosynthesis, melting of ice, cooking of an egg, decomposition (mostly), dissolving ammonium chloride/nitrate, sublimation.
- Exothermic examples: Combustion, combination, respiration, rusting of iron, neutralisation (mostly), decomposition of vegetable waste.