Decomposition reaction:
A decomposition reaction is a chemical reaction in which a single reactant breaks down into two or more simpler products under suitable conditions such as current, heat, and light.

Decomposition reaction
Types of decomposition reaction:
1. Thermal decomposition (by heat):
A decomposition reaction in which a compound breaks down into simpler substances when heat energy is supplied.
i. Decomposition of lead nitrate
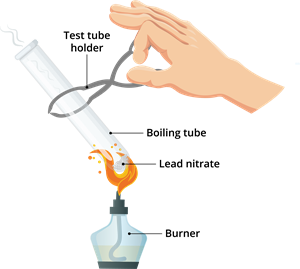
Decomposition of lead nitrate
- Lead nitrate (\(Pb(NO_3)_2\)): White crystalline solid
- Lead oxide (\(PbO\)): Yellow residue
- Nitrogen dioxide (\(NO_2\)): Brown fumes
- Oxygen gas (\(O_2\)): Colourless gas that supports combustion
ii. Decomposition of calcium carbonate
Calcium carbonate decomposes to produce calcium oxide and carbon dioxide on heating.
\(CaCO_3\) \(CaO + CO_2\)
- Calcium oxide (\(CaO\)) is called quick lime.
- Carbon dioxide gas is released.
- Used in the cement industry.
iii. Decomposition of ferrous sulphate
Ferrous sulphate decomposes to ferric oxide (\(Fe_2O_3\)), sulphur dioxide (\(SO_2\)) and sulphur trioxide (\(SO_3\)).
\(2FeSO_4\)(s) \(Fe_2O_3\)(s) + \(SO_2\)(g) + \(SO_3\)(g)
- The colour of the crystals changed from green to brown on heating.
- The brown solid formed is ferric oxide.
- The pungent smelling gas evolved is sulphur dioxide.
iv. Decomposition of ammonium chloride
Ammonium chloride decomposes to produce ammonia and hydrogen chloride gases.
\(NH_4Cl\)(s) \(NH_3\)(g) + \(HCl\)(g)
This reaction is reversible. On cooling the gases recombine to form ammonium chloride.
2. Electrolytic decomposition (by electricity):
A decomposition reaction in which a compound breaks down into simpler substances when electric current is passed through it.
The process in which water is decomposed by passing electricity is called the electrolysis of water.
\(2H_2O\)(l) \(2H_2\)(g) + \(O_2\)(g)
- Gas liberated at the cathode (hydrogen) and the anode (oxygen)
- Confirmation test: Hydrogen (pop sound) and oxygen (glows brightly)
- Volume ratio: \(H_2 : O_2\) \(=2:1\)
- A few drops of \(H_2SO_4\) is added to increase conductivity.
3. Photolytic decomposition (by light):
A decomposition reaction in which a compound breaks down into simpler substances when light energy (usually sunlight or UV light) is absorbed.
i. Decomposition of silver chloride:
The colour of white silver chloride changes to grey due to the decomposition of silver chloride under sunlight. This grey powder is silver, and chlorine gas is evolved.
ii. Decomposition of silver bromide:
The colour of silver bromide changes from pale yellow to greyish white due to the decomposition of silver bromide under sunlight. The grey powder is silver, and the red-brown bromine gas is evolved.
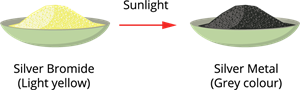
Photolytic decomposition of silver bromide
Application: Used in black and white photography.
Note: Most decomposition reactions are endothermic as they require energy to break down compounds. However, the decomposition of organic or vegetable waste into compost is an exothermic process, because heat is relesed during the microbial activity.