Displacement reaction:
A more reactive element displaces a less reactive element from its compound. It is also called as a single displacement reaction.
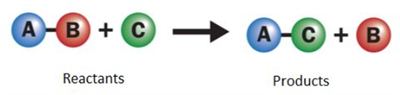
Single displacement reaction
Example:
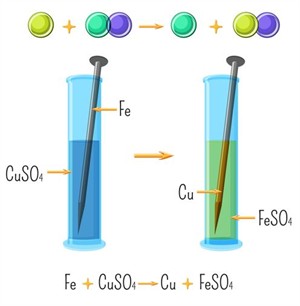
Reaction between iron and copper sulphate
- The Blue colour of copper sulphate solution fades and a pale green colour iron sulphate solution is formed.
- Brown coating forms on the nail.
- Iron is more reactive and displaces copper.
Double displacement reaction:
A double displacement reaction involves the exchange of ions between the reactants to form new products. A Double displacement reaction has the following two types:
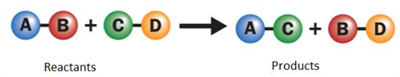
Types of double displacement reaction:
i. Neutralisation reaction:
Acid + Base → Salt + Water

Neutralisation reaction
ii. Precipitation reaction:
1.
Reactants: Both are colourless solutions
Products: White precipitate of (\(BaSO_4\))
2. \(Pb(NO_3)_2(aq) + 2KI(aq) → PbI_2(s) + 2KNO_3(aq)\)
Reactants: Both are colourless solutions
Products: Yellow precipitate of \(PbI_2\)
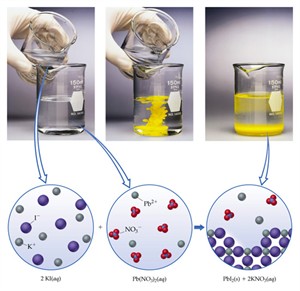
Precipitation of lead iodide
Redox reaction:
| Oxidation | Reduction |
| Gain of oxygen | Loss of oxygen |
| Loss of hydrogen | Gain of hydrogen |
| Loss of electrons | Gain of electrons |
| Increase in oxidation number | Decrease in oxidation number |
Redox: Both oxidation and reduction occur simultaneously.
Example:

Redox reaction
- Heat copper powder → turns black (\(CuO\))
- Pass hydrogen → turns brown again (\(Cu\)) metal
- Copper oxide gets reduced
- Hydrogen gets oxidised
| Agent | What does it do? | What happens to it? | Example |
| Oxidising agent | Causes oxidation of another substance | It gets reduced |
In \(CuO + H_2 → Cu + H_2O\),
\(CuO\) is the oxidising agent
|
| Reducing agent | Causes reduction of another substance | It gets oxidised |
In \(CuO + H_2 → Cu + H_2O\),
\(H_2\) is the reducing agent
|
Displacement and redox reactions play an important role in our daily lives, such as rusting, metal extraction etc. In the next session, we will explore a few more redox reactions, along with important real-life applications like corrosion and alloys, to understand how chemistry helps in protecting materials and improving their usefulness.