A displacement reaction is a chemical reaction in which atoms or ions of one element replace those of another element in a compound.
Displacement reactions are classified as:
- Single displacement reaction
- Double displacement reaction
i. Single displacement reaction:
A reaction in which more reactive element displaces a less reactive element from its compound.
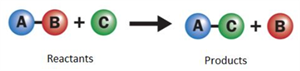
Single displacement reaction
Example: Iron nail placed in copper sulphate solution.
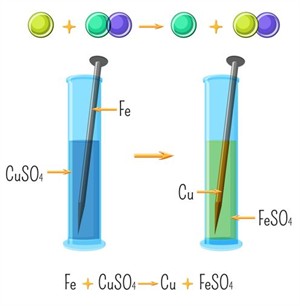
Reaction between iron and copper sulphate
Observation:
- The Blue colour of copper sulphate solution fades and a pale green colour iron sulphate solution is formed.
- Brown coating forms on the nail.
- Iron is more reactive and displaces copper.
Condition for the single displacement reaction to proceed: Only a more reactive element or metal can displace a less reactive element or metal. A less reactive metal cannot displace a more reactive one.
Reactivity series:
The reactivity series is a list of metals arranged from most reactive to least reactive.

Reactivity series
To remember the reactivity series:
"Please Stop Calling Me A Zebra, Instead Try Learning How Copper Saves Gold"
Observation of single displacement reaction:
Certain visible changes are observed in the reaction:
- Colour change of solution
- Deposition of metal
| Metal added | Metal displaced | Initial colour(solution) | Final colour (Solution) | Deposition |
| Iron (\(Fe\)) | Copper (\(Cu\)) | Blue | Green | Reddish-brown deposit forms |
| Zinc (\(Zn\)) | Copper (\(Cu\)) | Blue | Colourless | Reddish-brown deposit forms |
| Copper (\(Cu\)) | Silver (\(Ag\)) | Colourless | Blue | Grey deposit forms |
| Iron (\(Fe\)) | Silver (\(Ag\)) | Colourless | Pale green | Grey deposit forms |
| Zinc (\(Zn\)) | Iron (\(Fe\)) | Green | Colourless | Grey deposit forms |
ii. Double displacement reaction:
A double displacement reaction involves the exchange of ions between the two compounds to form two new compounds.
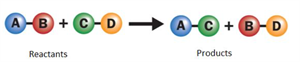
Double displacement reaction
Types of double displacement reaction:
A Double displacement reaction has the following two types:
i. Neutralisation reaction:
The reaction between an acid and a base result in the formation of salt and water is called a neutralisation reaction. This is another type of displacement reaction in which both acid and base neutralise each other.
Acid + Base → Salt + Water
Example: A common neutralisation reaction is the reaction of sodium hydroxide with hydrochloric acid. Here, sodium replaces hydrogen from hydrochloric acid, forming sodium chloride salt.

Neutralisation reaction
ii. Precipitation reaction:
When aqueous solutions of two compounds are mixed, if they react to form an insoluble compound and a soluble compound, it is called a precipitation reaction.
1.
Reactants: Both are colourless solutions
Products: White precipitate of (\(BaSO_4\))
2. \(Pb(NO_3)_2(aq) + 2KI(aq) → PbI_2(s) + 2KNO_3(aq)\)
Reactants: Both are colourless solutions
Products: Yellow precipitate of \(PbI_2\)
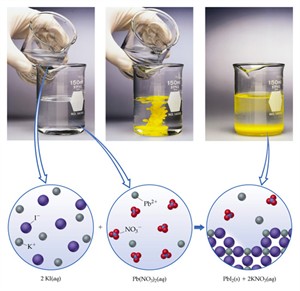
Precipitation of lead iodide