Have you ever wondered why milk turns sour if left outside, why iron objects rust, or how food gets cooked and digested? All these everyday changes are not random; they result from chemical reactions happening around us all the time.
Previously, we learned about the basics of chemistry and different types of changes in matter. In this chapter, we will strengthen that foundation by understanding chemical reactions, chemical equations, and how to represent them symbolically.
What is a reactant?
A substance participating in a chemical reaction, or a substance(s) left of the arrow in a chemical equation, is called a reactant.
What is a product?
Formed substance(s) in a chemical reaction or (a substance(s) to the right of the arrow in a chemical equation) are called products.
What is a chemical reaction?
A chemical reaction is a process in which one or more substances (reactants) are converted into new substances (products) with different properties.
Chemical reactions in daily life:
Chemical reactions are not limited to laboratories; they occur constantly around us. Consider the following situations:
-
Milk kept at room temperature during the summer
-
An iron nail left in a humid environment
-
Cooking of food
-
Digestion of food
-
Respiration
In all these cases, the original substances change in nature and identity, indicating that chemical reactions have occurred. Whenever a chemical change occurs, a chemical reaction has occurred.
Identification of a chemical reaction:
A chemical reaction can be identified by one or more of the following changes:
- Change in colour
- Change in state
- Change in temperature
- Evolution of gas
- Formation of precipitate
Burning of magnesium ribbon:
-
Magnesium ribbon burns with a dazzling white flame.
-
A white powder is formed.

Burning of magnesium ribbon
\(2Mg + O_2 → 2MgO\)
When magnesium oxide is dissolved in water, it turns red litmus paper blue, indicating that it is basic.
\(MgO + H_2O → Mg(OH)_2\)
This activity shows that a change in state and formation of a new substance indicate a chemical reaction.
Evolution of gas and the change in temperature:
-
Bubbles of hydrogen gas are evolved.
-
The test tube becomes hot, indicating a temperature rise.
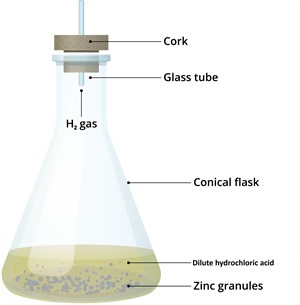
Reaction between zinc and hydrochloric acid
\(Zn + 2HCl → ZnCl_2 + H_2\)
The evolution of gas and the change in temperature confirm that a chemical reaction has taken place.
Chemical equations:
To understand chemical reactions more clearly and represent them precisely and scientifically, scientists use chemical equations. A chemical equation is the symbolic representation of a chemical reaction using chemical formulae.
Writing a chemical equation:
- Reactants: LHS
- Products: RHS
- Right arrow (→): Direction reaction by pointing to the products
- Physical states: Solid – (s), Liquid – (l), Gas – (g), Aqueous – (aq)
Law of conservation of mass:
According to the law of conservation of mass, mass can neither be created nor destroyed in a chemical reaction.
Therefore, the number of atoms of each element must be the same on both sides of a chemical equation.
Therefore, the number of atoms of each element must be the same on both sides of a chemical equation.
Word equation:
Iron + Water → Iron oxide + hydrogen
Skeletal chemical equation:
\(Fe + H_2O → Fe_3O_4 + H_2\)
This equation is unbalanced because the number of atoms on both sides is not equal.
Balancing chemical equations (Hit and trial method):
This method involves adjusting coefficients to ensure that atoms are balanced on both sides using the smallest whole numbers.
\(Fe + H_2O → Fe_3O_4 + H_2\)
Steps to balance:
- Count the atoms of each element on both sides .
| Element |
Number of atoms in reactants (LHS)
|
Number of atoms in products (RHS)
|
|
\(Fe\)
|
\(1\)
|
\(3\)
|
|
\(H\)
|
\(2\)
|
\(2\)
|
|
\(O\)
|
\(1\)
|
\(4\)
|
- Add coefficients to balance
- Check for correctness
- Include physical states
Balanced equation:
\(3Fe({s}) + 4H_2O({g}) → Fe_3O_4({s}) + 4H_2({g})\)
Reaction conditions like temperature, pressure, or catalysts may be written above or below the arrow.
Chemical reactions help us understand how substances change and interact. In the next section, we will explore the different types of chemical reactions and discover how substances combine or break apart in fascinating ways.