In previous session, we learned about chemical reactions, balancing chemical equations, and the law of conservation of mass. We understood that during a chemical reaction, atoms are neither created nor destroyed; instead, bonds between atoms break and new bonds are formed to produce new substances.
Classification of chemical reactions:
Chemical reactions are classified based on the nature of reactants and products. The major types are:
-
Combination reactions
-
Decomposition reactions
-
Displacement reactions
-
Double displacement reactions
-
Oxidation and reduction reactions
In this session, we will focus on the combination reaction.
Combination reactions:
A combination reaction occurs when two or more reactants combine to form one product. It is also referred to as a synthesis reaction .

Combination reaction
Examples of combination reaction:
i. Formation of slaked lime:
When calcium oxide (quick lime) reacts with water, calcium hydroxide (slaked lime) is formed with the release of heat (exothermic).
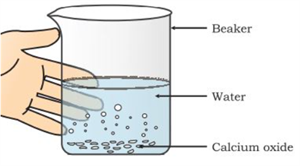
Formation of calcium hydroxide
Observation:
-
A hissing sound is produced
-
The beaker becomes hot
-
Heat is released (exothermic reaction)
\(CaO(s) + H_2O(l) → Ca(OH)_2(aq)\)
Application: Whitewashing of walls
The slaked lime formed reacts slowly with carbon dioxide present in air:
\(Ca(OH)_2 + CO_2 → CaCO_3 + H_2O\)
The formation of calcium carbonate gives walls a shiny and durable finish. Interestingly, marble has the same chemical composition
ii. Burning of coal:
\(C(s) + O_2(g) → CO_2(g)\)
Coal (carbon) burns in the air to form carbon dioxide gas.
Combustion reactions as examples of combination reactions.
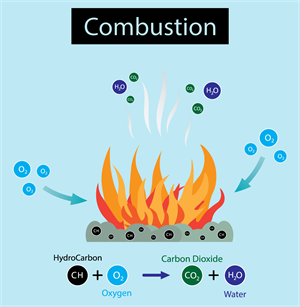
Combustion reaction
iii. Formation of water:
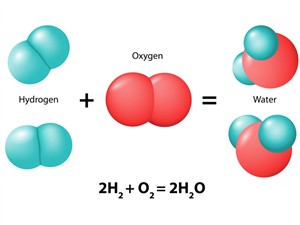
Formation of water
\(2H_2(g) + O_2(g) → 2H_2O(l)\)
In the presence of oxygen, hydrogen burns to produce water in the form of steam. Upon cooling, it becomes liquid.