Combination reactions:
A combination reaction occurs when two or more reactants combine to form one product. It is also referred to as a synthesis reaction .

Combination reaction
Examples of combination reaction:
i. Formation of slaked lime:
When calcium oxide (quick lime) reacts with water, calcium hydroxide (slaked lime) is formed with the release of heat (exothermic).
Observation:
-
A hissing sound is produced
-
The beaker becomes hot
-
Heat is released (exothermic reaction)
\(CaO(s) + H_2O(l) → Ca(OH)_2(aq)\)
Application: Whitewashing of walls
The slaked lime formed reacts slowly with carbon dioxide present in air:
\(Ca(OH)_2 + CO_2 → CaCO_3 + H_2O\)
The formation of calcium carbonate gives walls a shiny and durable finish. Interestingly, marble has the same chemical composition
ii. Burning of coal:
\(C(s) + O_2(g) → CO_2(g)\)
Coal (carbon) burns in the air to form carbon dioxide gas.
Combustion reactions as examples of combination reactions.
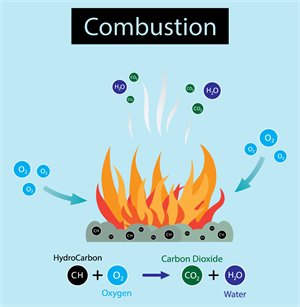
Combustion reaction
Decomposition reaction:
A decomposition reaction is a chemical reaction in which a single reactant breaks down into two or more simpler products under suitable conditions such as current, heat, and light.

Decomposition reaction
Types of decomposition reaction:
1. Thermal decomposition (by heat):
A decomposition reaction in which a compound breaks down into simpler substances when heat energy is supplied.
i. Decomposition of lead nitrate
- Lead nitrate (\(Pb(NO_3)_2\)): White crystalline solid
- Lead oxide (\(PbO\)): Yellow residue
- Nitrogen dioxide (\(NO_2\)): Brown fumes
- Oxygen gas (\(O_2\)): Colourless gas that supports combustion
ii. Decomposition of calcium carbonate
Calcium carbonate decomposes to produce calcium oxide and carbon dioxide on heating.
\(CaCO_3\) \(CaO + CO_2\)
- Calcium oxide (\(CaO\)) is called quick lime.
- Carbon dioxide gas is released.
- Used in the cement industry.
iii. Decomposition of ferrous sulphate
Ferrous sulphate decomposes to ferric oxide (\(Fe_2O_3\)), sulphur dioxide (\(SO_2\)) and sulphur trioxide (\(SO_3\)).
\(2FeSO_4\)(s) \(Fe_2O_3\)(s) + \(SO_2\)(g) + \(SO_3\)(g)
- The colour of the crystals changed from green to brown on heating.
- The brown solid formed is ferric oxide.
- The pungent smelling gas evolved is sulphur dioxide.
2. Electrolytic decomposition (by electricity):
A decomposition reaction in which a compound breaks down into simpler substances when electric current is passed through it.
The process in which water is decomposed by passing electricity is called the electrolysis of water.
\(2H_2O\)\((l)\) \(2H_2\)\((g)\) + \(O_2\)\((g)\)
- Gas liberated at the cathode (hydrogen) and the anode (oxygen)
- Confirmation test: Hydrogen (pop sound) and oxygen (glows brightly)
- Volume ratio: \(H_2 : O_2\) \(=2:1\)
- A few drops of \(H_2SO_4\) is added to increase conductivity.
3. Photolytic decomposition (by light):
A decomposition reaction in which a compound breaks down into simpler substances when light energy (usually sunlight or UV light) is absorbed.
i. Decomposition of silver chloride:
The colour of white silver chloride changes to grey due to the decomposition of silver chloride under sunlight. This grey powder is silver, and chlorine gas is evolved.
ii. Decomposition of silver bromide:
The colour of silver bromide changes from pale yellow to greyish white due to the decomposition of silver bromide under sunlight. The grey powder is silver, and the red-brown bromine gas is evolved.
Application: Used in black and white photography.
Note: Most decomposition reactions are endothermic as they require energy to break down compounds. However, the decomposition of organic or vegetable waste into compost is an exothermic process, because heat is relesed during the microbial activity.
Displacement reaction:
A displacement reaction is a chemical reaction in which atoms or ions of one element replace those of another element in a compound.
Displacement reactions are classified as:
- Single displacement reaction
- Double displacement reaction
i. Single displacement reaction:
A reaction in which more reactive element displaces a less reactive element from its compound.
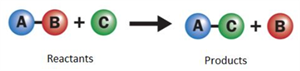
Single displacement reaction
Example: Iron nail placed in copper sulphate solution.
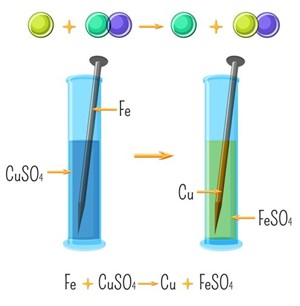
Reaction between iron and copper sulphate
Observation:
- The Blue colour of copper sulphate solution fades and a pale green colour iron sulphate solution is formed.
- Brown coating forms on the nail.
- Iron is more reactive and displaces copper.
Condition for the single displacement reaction to proceed: Only a more reactive element or metal can displace a less reactive element or metal. A less reactive metal cannot displace a more reactive one.
Reactivity series:
The reactivity series is a list of metals arranged from most reactive to least reactive.

Reactivity series
To remember the reactivity series:
"Please Stop Calling Me A Zebra, Instead Try Learning How Copper Saves Gold"
Observation of single displacement reaction:
Certain visible changes are observed in the reaction:
- Colour change of solution
- Deposition of metal
ii. Double displacement reaction:
A double displacement reaction involves the exchange of ions between the two compounds to form two new compounds.
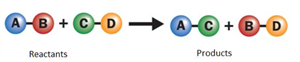
Double displacement reaction
Types of double displacement reaction:
A Double displacement reaction has the following two types:
i. Neutralisation reaction:
The reaction between an acid and a base result in the formation of salt and water is called a neutralisation reaction. This is another type of displacement reaction in which both acid and base neutralise each other.
Acid + Base → Salt + Water
Example: A common neutralisation reaction is the reaction of sodium hydroxide with hydrochloric acid. Here, sodium replaces hydrogen from hydrochloric acid, forming sodium chloride salt.

Neutralisation reaction
ii. Precipitation reaction:
When aqueous solutions of two compounds are mixed, if they react to form an insoluble compound and a soluble compound, it is called a precipitation reaction.
1.
Reactants: Both are colourless solutions
Products: White precipitate of (\(BaSO_4\))
2. \(Pb(NO_3)_2(aq) + 2KI(aq) → PbI_2(s) + 2KNO_3(aq)\)
Reactants: Both are colourless solutions
Products: Yellow precipitate of \(PbI_2\)
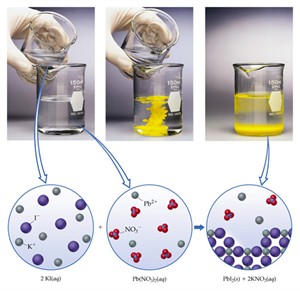
Precipitation of lead iodide