Types of chemical reactions of organic compounds:
-
Combustion: Combustion is burning of carbon compounds in air to form \(CO_2\), \(H_2O\), heat, and light. It is a type of oxidation reaction (addition of oxygen).
-
Blue vs Yellow flame:Complete combustion (sufficient oxygen, saturated compounds) - Blue flameIncomplete combustion (less oxygen, unsaturated compounds) - Yellow, sooty flame
-
Oxidation: Ethanol to Ethanoic acid using alkaline \(KMnO_4\)
-
Addition: Unsaturated hydrocarbons (ethene) + hydrogen to saturated hydrocarbons (ethane)
-
Substitution: Methane reacts with chlorine in sunlight to form chloromethane
Ethanol (\(C_2H_5OH\)):
Ethanol, commonly referred to as alcohol, is a simple organic compound with the molecular formula \(C_2H_5OH\). It is a colourless, volatile liquid with a pleasant odour and a burning taste. Found in alcoholic beverages and medicinal syrups, ethanol holds significant industrial and domestic importance.
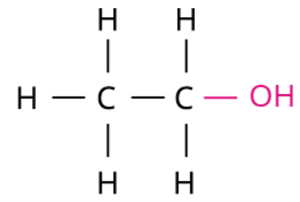
Skeletal structure of ethanol
Chemical properties of ethanol (\(C_2H_5OH\)):
i. Dehydration to ethene: \(CH_3CH_2OH \xrightarrow [443K]{Conc H_2SO_4} CH_2=CH_2 + H_2O\)
ii. Reaction with sodium: \(2CH_3CH_2OH + 2Na \rightarrow 2C_2H_5ONa + H_2\)
iii. Oxidation:
iv. Dehydrogenation: \(CH_3CH_2OH \xrightarrow [573K]{Cu} CH_3CHO + H_2\)
v. Esterification: \(CH_3CH_2OH + CH_3COOH \xrightarrow {Conc H_2SO_4} CH_3COOC_2H_5 + H_2O\)
Ethanoic acid (\(CH_3COOH\)):
Ethanoic acid is a key carboxylic acid, widely used in food and industry. When pure, it forms ice-like crystals and is called glacial acetic acid.
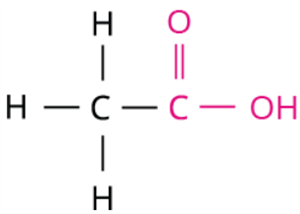
Skeletal structure of ethanoic acid
Chemical properties of ethanoic acid:
i. Reaction with metal: \(2CH_3COOH + 2Na \rightarrow 2CH_3COONa + H_2\)
ii. Reaction with carbonates and bicarbonates: \(2CH_3COOH + NaCO_3 \rightarrow 2CH_3COONa + CO_2 + H_2O\)
iii. Reaction with base: \(CH_3COOH + NaOH \rightarrow CH_3COONa + H_2O\)
iv. Decarboxylation (removal of \(CO_2\)): \(CH_3COOH \xrightarrow {NaOH /CaO} CH_4+ Na_2CO_3\)
PYQ - Carbon compounds, Conversion
Soaps:
Soaps are sodium or potassium salts of long-chain carboxylic acids, called fatty acids. Soap generally requires the use of two major raw materials:
- Fat
- Alkali
Sodium hydroxide is the most common alkali used in soap production. Potassium hydroxide is yet another alternative. A potassium-based soap is far more water-soluble than a sodium-based soap.
Based on the above features, there are two types of soaps:
- Hard soap: Soaps made by saponifying oils or fats with caustic soda (sodium hydroxide) are referred to as hard soaps. They are typically used for washing.
- Soft soap: Soaps that are made by saponifying oils or fats with potassium salts are referred to as soft soaps. They are employed in the cleansing of the body.
The alkaline hydrolysis of esters (using alkali like sodium hydroxide) is known as saponification. The chemical equation of saponification is
\(CH_3COOCH_3 + NaOH → CH_3COONa + CH_3OH\)
Cleansing action of soap:
- A soap molecule is made up of two chemically distinct parts that interact with water in different ways. It has one polar end with a short head carboxylate group (\(-COONa\)) and one non-polar end with a long tail made of the hydrocarbon chain.
- The polar end is hydrophilic (water-loving) in nature, and it is drawn to water. The non-polar end is hydrophobic (hates water) in nature, and it is attracted to dirt or oil on the cloth but not to water. As a result, the hydrophobic part of the soap molecule traps the dirt while the hydrophilic part makes the entire molecule water-soluble.
-
When soap or detergent is dissolved in water, the molecules form clusters known as 'micelles'. Their long hydrocarbon chains bind to the oil and dirt. As a result, the dirt is surrounded by the non-polar end of the soap molecules. The micelles are water-soluble because of the charged carboxylate end of the soap molecules. As a result, the soap washes away the dirt.
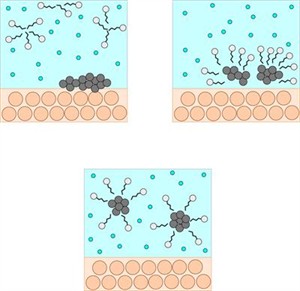
Cleansing action of soap
Effect of hard water on soap and rise of detergents:
The presence of calcium and magnesium ions (\(Ca^{2+}\) and \(Mg^{2+}\)) in hard water limits the cleaning action of soap. When combined with soap, hard water forms a thin layer (precipitates of metal ions) known as scum, which leaves a deposit on the clothes or skin and is difficult to remove. This can cause the fabric to deteriorate over time and eventually ruin the clothes. To overcome this problem, synthetic detergents were developed.
Unlike soaps (which are sodium salts of fatty acids), detergents are usually sodium salts of sulphonic acids or alkyl hydrogen sulphates. These do not form scum with hard water ions, allowing detergents to maintain their cleaning action in both hard and acidic water.
Difference between soaps and detergents:
|
Soap
|
Detergent
|
| The sodium salt of long-chain fatty acids makes up the soap. | Sodium salts of sulphonic acids make up the detergent. |
| \(-COO^-Na^+\) is the ionic part of the soap. | \(-SO_3^-Na^+\) is the ionic part of the detergent. |
| Soap is manufactured from animal fats or vegetable oils. | Detergents are obtained from hydrocarbons extracted from crude oil. |
| The effectiveness of soap reduces in hard water. | Detergents are effective in hard water. |
| Soap forms a scum in hard water. | Detergents do not form scum in hard water. |
| Soap has a poor foaming capacity. | Detergents have a rich foaming capacity. |
| They are biodegradable. | They are mostly non-biodegradable. |
Have you ever noticed the term "TFM" mentioned on the soap cover?:
What is TFM in Soaps?
TFM (Total Fatty Matter) indicates the quality of a soap. According to Indian standards:
-
Bathing bars: TFM between 40% and 60%
-
Toilet soaps: TFM between 60% and 76%
Higher TFM means better moisturising, gentler cleansing, and overall superior soap quality.
PYQ - Soaps, Saponification reaction
So next time you see those stubborn stains disappear or your shampoo lather up, remember it’s all clever molecules working behind the scenes. Understanding soaps and detergents isn’t just about cleaning, it’s about unlocking the science in everyday magic.