Ionic compounds (Transfer of electrons):
Metal + non-metal → Ionic bond (or) electrovalent bond
Metal: Loss of electron - Cation (\(+\))
Non-metal: Gain of electron - Anion (\(-\))
Non-metal: Gain of electron - Anion (\(-\))
Examples: \(NaCl\), \(MgCl_2\), \(Na_2O\), \(MgO\) and \(AlCl_3\)
Formation of \(MgCl_2\):
- Magnesium atom: Electrons present = \(12\), Electronic configuration = (\(2, 8, 2\))
- Chlorine atom: Electrons present = \(17\), Electronic configuration = (\(2, 8, 7\))
- Cation: \(Mg^{2+}\)
- Anion: \(Cl^-\)
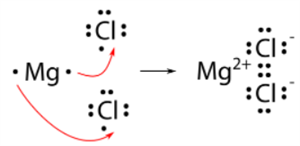
Magnesium chloride formation
PYQ - Ionic compounds
Properties of ionic compounds:
- Physical state: Crystalline solids. Strong force of attraction between ions.
- High melting and boiling points: Large amount of energy is required to break inter-ionic forces
- Solubility: Soluble in polar solvents (water); Insoluble in non-polar solvents
- Conductivity: Do not conduct electricity in a solid state → ions are fixed. Conduct in molten state or aqueous solution → ions become free to move.
Occurrence of metals:
High reactive metals (\(K, Na, Ca, Mg, Al\))
-
Never found free
-
Strong affinity for oxygen
-
Extracted by electrolysis
Moderate reactive metals (\(Zn, Fe, Pb, Cu\))
-
Found as oxides, sulphides, carbonates
-
Extracted by roasting/calcination \(+\) reduction
Low reactive metals (Ag, Au, Pt)
-
Found native (free)
-
Often require simple heating
Ores extracted from the earth usually contain large amounts of impurities such as soil, sand, etc., called gangue. Impurities must be removed from the ore before the metal can be extracted.
PYQ - Metal and its compounds
Extraction of metals based on reactivity:
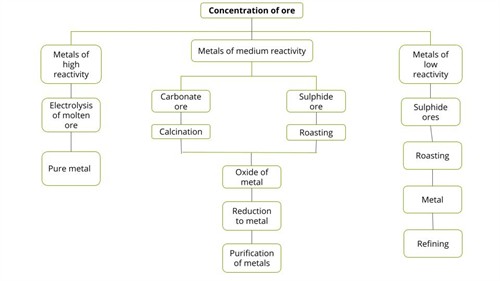
Extraction of metals
PYQ - Extraction of metals, Extraction of mercury, Electrolytic reduction, Conversion of ores to metals
Thermit reaction:
\(Fe_2O_3(s) +2Al(s) → 2Fe(l) + Al_2O_3(s) +\ heat\)
Type of reaction: Displacement and exothermic
Application: Joining railway tracks, cracked machine parts and welding
Electrolytic refining:
Copper, zinc, tin, nickel, silver, gold and other metals are purified electrolytically.
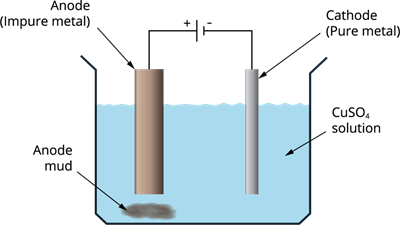
Refining of metals
Corrosion:
Corrosion is the progressive destruction of metals by the action of air, moisture or chemicals (such as an acid) on their surface.
| Metal | Reaction | Colour of the product |
| Silver | \(Ag + S → Ag_2S\) | Black (Tarnish) |
| Copper | \(Cu + H_2O + CO_2 → CuCO_3\) | Green (Patina) |
| Iron | \(Fe + O_2 + H_2O → 2Fe_2O_3.xH_2O\) | Reddish brown (Rust) |
Prevention of corrosion:
-
Painting
-
Oiling/greasing
-
Galvanisation (Zinc coating)
-
Chrome plating
-
Anodising
-
Alloying
PYQ - Corrosion
Alloys:
A homogeneous mixture of metals (or metals + non-metals) to improve properties.
| Alloy | Composition | Uses |
| Brass | \(Cu + Zn\) | Decorative items, instruments |
| Bronze | \(Cu + Sn\) | Statues, tools and medals |
| Solder | \(Pb + Sn\) | Joining electrical components, plumbing etc |
| Amalgam | \(Metals + Hg\) | Dental fillings |
| Stainless Steel | \(Fe + Cr + Ni\) | Utensils, medical tools |
| Gold Alloy |
\(Au + Cu/Ag\)
22 parts gold + 2 parts Cu/Ag
|
Harder than pure gold; used in jewellery |
PYQ - Metal and its alloys