In the previous session, we observed the properties of acids and bases. We also learned that some acids and bases can be corrosive, meaning they can damage materials and harm our skin.
So, how can we safely identify whether a substance is acidic or basic without touching or tasting it?
To solve this problem, scientists use special substances called indicators.
Acid–Base Indicators:
The chemical substance that changes colour depending on the pH of the solution, indicating whether the solution is acidic, basic or neutral, is called an acid-base indicator.
Types of Indicators:
Indicators are mainly classified into two types:
i. Natural indicators
These are obtained from natural sources like plants and microorganisms.
Examples: Litmus, turmeric, china rose.
Examples: Litmus, turmeric, china rose.
ii. Synthetic indicators
These are man-made indicators prepared in laboratories.
Examples: Phenolphthalein, methyl orange.
Examples: Phenolphthalein, methyl orange.
In this session, we will learn in detail about one of the most commonly used natural indicators, litmus.
Litmus as an indicator:
Can you believe that a piece of paper helps you to figure out whether it is an acid or a base when it just takes a slight dip in liquids? Yes, a special paper called Litmus.
Litmus paper is made from lichens, which are tiny plant-like organisms made of two separate organisms, a fungus and an alga. This partnership is helping with solution detection. And we use it mostly in the form of litmus paper, which is available in blue, red, and purple colours.

Lichens
Activity: Testing solutions with litmus paper
Materials Required:
-
Red and blue litmus paper
-
Lemon Juice
-
Vinegar
-
Soap solution
-
Baking soda solution
-
Lime water
-
Distilled water
-
Sugar solution
Instructions:
Step 1: Take the strips of red litmus paper.
Step 2: Take the sample solutions in separate test tubes and dip a red litmus paper into each test tube and observe the colour change.
Step 3: Repeat the same with the strips of blue litmus paper and observe the colour change.
Observation: The observations are tabulated as follows.
|
Samples
|
Red litmus
|
|
No change
|
|
|
No change
|
|
|
Blue
|
|
|
Blue
|
|
|
No change
|

Litmus test: Acid, base and neutral solution
Result:
- If the liquid is sour and fizzy, it detects an acid, and the blue strip turns red.
- If the liquid is bitter and slippery, it detects a base, and the red strip turns blue.
- If the liquid is calm and chilled, it detects neutral, and both red and blue litmus remain unchanged.
| Litmus paper | Test with the acid substance | Test with the base substance |
| Blue litmus paper |
Blue \(→\) Red
|
Blue \(→\) Blue (No change)
|
| Red litmus paper |
Red \(→\) Red (No change)
|
Red \(→\) Blue
|
Many students often confuse the term “lime” with the fruit lemon or lime used in food. However, in science, lime refers to a chemical substance.
Limewater is a solution of calcium hydroxide in water and is commonly used in experiments. It can be prepared in the laboratory using the following steps:
Preparation of limewater:
i. Take a small amount of calcium oxide (quicklime) in a clean beaker.
ii. Carefully add water to it and stir well, calcium oxide reacts with water to form calcium hydroxide.
\(CaO + H_2O → Ca(OH)_2 + Heat\)
iii. Leave the mixture undisturbed for about an hour so that the solid particles can settle down.
iv. After some time, a clear liquid will be seen above the settled solid.
v. Filter this clear liquid into another container using filter paper.
vi. The clear solution obtained is called limewater, which is a solution of calcium hydroxide in water.
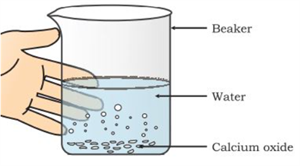
Formation of calcium hydroxide