Imagine the process of constructing a house. The initial step in this process involves gathering essential raw materials, which serve as the foundation of the entire structure. Key materials such as cement, bricks, and iron rods are crucial to ensuring the strength and stability of the house. Without these fundamental building blocks, the dream of a completed house would remain unattainable.

Construction of a building
In a broader context, this concept applies to everything in the universe around us. Just as a house is built from specific materials, all matter is composed of basic building blocks known as elements. These elements are the fundamental substances that combine in various ways to create the diverse array of materials and compounds that shape our world.
Let us learn in detail about the elements and their classification.
Element
The simplest form of matter is an element. Each element is made up of identical particles called atom. Atoms are the simplest particles of an element that cannot be broken down in a chemical reaction.
The term element was first used by Robert Boyle. He was a supporter of the elemental essence of matter and the nature of the vacuum. Boyle's Law was his best-known work.
According to Antoine-Laurent Lavoisier, an element is a fundamental type of matter that cannot be broken down into simpler substances by chemical reactions. A chemical element is a material that cannot be broken down into simpler forms using traditional chemical methods.
All the elements in the periodic table are example for elements - such as oxygen, iron, gold, boron, sulphur, etc.
An atom is the smallest unit of a chemical element. Atoms are incredibly tiny, smaller than anything we can imagine or compare them to.
Subatomic particles (sub- means "smaller size") make up an atom. These particles are the
- Proton (\(p^+\)), a positively (\(+\)) charged particle.
- Electron (\(e^–\)), a negatively (\(–\)) charged particle.
- Neutron (\(n^0\)), a neutral particle with no charge (\(0\)).
All the atoms in the periodic table are pure substances and are called elements. The periodic table consists of \(118\) elements. Some of the elements and their symbols are listed below,
|
Element
|
Symbol
|
Element
|
Symbol
|
|
Oxygen
|
O |
Calcium
|
Ca |
|
Carbon
|
C |
Aluminium
|
Al |
|
Sodium
|
Na |
Sulphur
|
S |
|
Hydrogen
|
H |
Boron
|
B |
|
Iron
|
Fe |
Magnesium
|
Mg |
|
Chlorine
|
Cl |
Copper
|
Cu |
|
Nitrogen
|
N
|
Lithium
|
Li |
Each element has different physical and chemical properties. All the atoms in the periodic table are pure substances and have different physical and chemical properties. Based on the physical and chemical properties, the elements are classified as,


- Metals
- Nom-metals
- Metalloids
1. Metals:
Metals are electropositive elements that donate electrons to form a stable configuration. A physical property can be observed and measured without altering the sample's chemical identity.
- Malleable (can be drawn into thin sheets)
- Ductile (can be drawn into wires)
- Lustre (shine) with silvery grey or golden yellow in colour
-
Sonorous (makes a ringing sound when hit)
-
Most metals are solids at room temperature, except mercury, which is a liquid at room temperature.

Properties of metals
-
All metals are hard except sodium and potassium.
-
Metals have \(1\) to \(3\) electrons in their outermost shell.
-
Metals are generally good conductors of heat and electricity (they have free electrons in their outermost shells).
-
Metals have a high density (mass per unit volume).
-
Metals have high melting and boiling points because of their strong metallic bonds, except for sodium and potassium, which have low melting and boiling points.
The materials which generally possess the above properties are called metals.
2. Non-metals:
Non-Metals are electronegative elements that gain electrons to form a stable configuration.
- Non-metals are non-ductile in nature (as they are very brittle, they cannot be drawn into wires).
- Non-metals are soft except diamond.

Properties of non-metals
- Non-metals are generally poor conductors of heat and electricity, except graphite, which conducts electricity.
- The materials which generally possess the above properties are called non-metals.
Example:
Oxygen, carbon, sulfur, hydrogen, phosphorus, nitrogen, halogens and noble gases.
3. Metalloids:
They show the characteristics of both metals and non-metals and are known as metalloids. There are around \(8\) elements in the periodic table that are called metalloids.
Example:
Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), Tellurium (Te), Polonium (Po).
Physical properties:
- Metalloids are solid at room temperature.
- They can form alloys with other metals.
- Some metalloids (silicon and germanium) can behave as electrical conductors under specific conditions. Hence, they are called semiconductors.
- Silicon appears lustrous; however, it is neither malleable nor ductile (it is brittle, a characteristic of some non-metals).
- The physical properties of metalloids tend to be metallic, but their chemical properties tend to be non-metallic.
Compounds:
Compounds are a form of matter created by combining two or more elements in a specific mass ratio. Chemical methods may be used to decompose it into its constituent components. It is a form of matter created by combining two or more elements in a specific mass ratio. To decompose it into its constituent components, we use chemical methods.
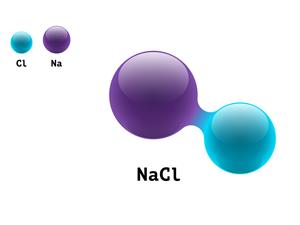
Sodium chloride
A chemical compound is formed when two or more distinct elements are chemically combined, i.e. chemical bonds form between their atoms.
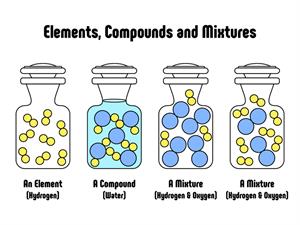
Pure substances and mixtures
Properties of Compounds:
- Two or more elements are chemically combined in a compound.
- The elements in a compound are present in a fixed mass ratio.
- Physical methods cannot isolate the constituents of a compound.
- The constituents of a compound lose their identities, i.e., a compound's properties vary from those of its constituent elements.
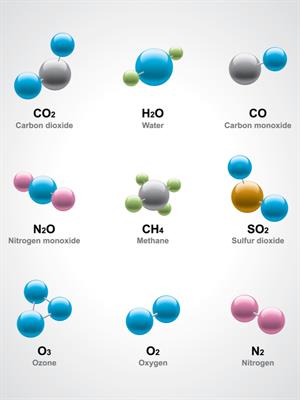
Compounds and molecules
Constituent elements in a compound:
- Water (\(H_2O\)) has three atoms: Two hydrogen (\(H\)) atoms and one oxygen (\(O\)) atom.
- Methane (\(CH_4\)) with five atoms: One carbon (\(C\)) atom and four hydrogen (H) atoms.
- Glucose (\(C_6H_{12}O_6\)) contains elements of carbon \(6\), hydrogen \(12\) and oxygen \(6\) combined to form a glucose molecule.
- Sodium chloride (\(NaCl\)) contains the two elements: sodium and chlorine atoms combined to form a sodium chloride molecule in a \(1:1\) ratio.
Activity: Comparing the nature of a mixture and a compound
Instructions:
Step 1: In a watch glass, take \(5.6 g\) of iron filings and \(3.2 g\) of sulfur powder. Mix them well in the watch glass and label this mixture as Sample \(A\).
Step 2: From Sample A, take half of the mixture and transfer it to a china dish. Heat the mixture, stirring constantly, until it turns black.
Step 3: Once the mixture has cooled, transfer it from the china dish to a mortar and grind it with a pestle. Finally, transfer the ground mixture to another watch glass and label it as Sample \(B\).
Observation:
i. Appearance:
Upon examining Sample \(A\), it is clear that the iron and sulfur components retain their distinct properties. The individual black and yellow particles of iron and sulfur are easily visible, indicating that this sample is a mixture of these elements.

In contrast, Sample \(B\) presents a uniform black mass, which is iron sulfide. Sample B has a uniform texture and colour throughout, resulting from the heating of iron and sulfur together. Therefore, Sample B is a compound (iron sulphide).

Sample B: Iron sulphide
The reaction is as given below,
\(\text{Iron} + \text{Sulfur} \to \text{Iron sulphide}\)
ii. Magnetic effect:
When a magnet is brought close to Sample \(A\), the iron filings in the mixture are attracted it. Indicating that iron and sulfur can be separated through physical means.

Magnetic separation on Sample A
In contrast, when a magnet is placed near Sample \(B\), it does not attract the components. This suggests that the compound has properties that are different from those of its component elements, iron and sulfur.

Magnetic separation on Sample B
ii. Gas test:
To a small amount of Sample \(A\) in a test tube, add a few drops of dilute hydrochloric acid. A gas will evolve, and when you bring a glowing splinter near the mouth of the test tube, the flame will produce a popping sound. The gas released is colourless and odourless, confirming the presence of hydrogen gas.
The reaction is as given below,
\(\text{Iron} + \text{dilute hydrochloric acid} \to \text{Iron chloride} + \text{Hydrogen gas}\)
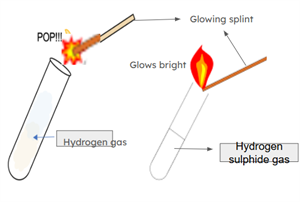
Gas test
Similarly, when you add a few drops of dilute hydrochloric acid to Sample \(B\) in another test tube, gas is released, and it has a rotten-egg smell. When a glowing splinter is placed near the mouth of this test tube, the flame continues to burn steadily.
\(\text{Iron sulphide} + \text{dilute hydrochloric acid} \to \text{Iron chloride} + \text{Hydrogen sulphide}\)
Thus, the above tests confirm that Sample A is a mixture of iron and sulfur, while Sample \(B\) is an iron sulfide compound; both exhibit distinct physical and chemical properties.