Surprisingly, the same types of particles make up everything around us: the book in your hand, the pencil you use, the air you breathe, the food you eat, and even the stars in the galaxy. Everything is made of matter!
If a rock and a butterfly are made of matter, why are they different? The incredible part of nature is that the arrangement of matter makes all the difference. The study of the nature of matter explores how these tiny, invisible particles combine to form the things around us. It reveals the secret behind our world and ourselves.
In this chapter, we will learn about the nature of matter and how elements, compounds, and mixtures combine to form the various materials we encounter every day.
Mixtures:
Consider a bowl of fruit salad, containing various types of fruit. These fruits can be distinguished by appearance and separated by handpicking. This is an example of a mixture.
In a mixture, substances are combined so that each retains its individual properties. The individual substances that are combined to make a mixture are known as components.
A mixture is a combination of two or more substances, composed of two or more elements, compounds, or both, that are physically combined.
Example: Oil and water, sand and water, and salads
The components of the mixture do not undergo any chemical reactions with each other. In some mixtures, the components are visible, while in others, they cannot be seen with the naked eye. Based on the visibility and nature of components, mixtures are classified as follows:
- Uniform
- Non-uniform
1. Uniform mixture:
- All components of the mixture blend evenly.
- There are no visible barriers; the mixture appears uniform.
- It is a single-phase solution.
- Example: Salt in water, sugar in water
- Uniform mixtures are also called homogeneous mixtures.
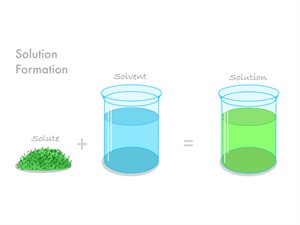
Uniform mixture
In a homogeneous mixture, the components of the solution cannot be seen with the naked eye.
2. Non-uniform mixture:
- All components of the mixture are unevenly distributed.
- There are noticeable separate lines.
- There are two or more phases in the solution.
- Oil and water, sand and water, salad are examples.
- Non-uniform mixtures are also called heterogeneous mixtures.
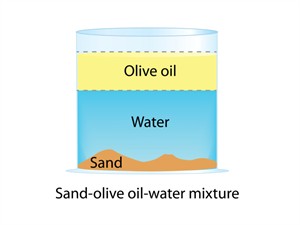
Non-uniform mixture
Have you ever wondered what air is made of? Is it a single substance or a combination of many components? Let us understand the composition of air.
Is air a mixture?
Air is a mixture. It is a combination of two or more substances that do not react chemically with each other. Air is a uniform mixture of gases, each retaining its properties. The main gases in the air are as follows:
- Nitrogen (\(~78%\))
- Oxygen (\(~21%\))
- Argon (\(~0.9%\))
- Carbon Dioxide (\(~0.04%\))
- Air also contains water vapour (moisture).
Activity: To test the presence of carbon dioxide in the air
Preparation of Lime Water:
- Take a test tube and fill it with water.
- Add a small amount of calcium oxide (quicklime) to the water.
Quick lime reacts vigorously with water, forming calcium hydroxide and releasing heat. The resulting solution is commonly known as lime water. Filter the solution, and it will appear as a colourless liquid.
Lime Water Turns Milky:
- Place the lime water in a test tube in an open space.
- Stir the solution at regular intervals.

A clear solution of lime water turns milky
You will observe that the lime water gradually turns milky white.
Reason:
When lime water is exposed to air, it reacts with the carbon dioxide present in the air and turns milky white (due to the formation of calcium carbonate). Calcium carbonate appears as insoluble, tiny white particles. Hence, the solutions turn milky white, indicating the following reaction,
Calcium hydroxide + Carbon dioxide \(\to\) Calcium carbonate + Water
Types of mixture:
Mixtures can be of various types based on the nature and physical state of their components. The types of mixtures based on their physical states are as follows:
- Solids in a solid - alloys
- Solids in a liquid - salt solution, sugar solution
- Liquids in a liquid - lemonade
- Gas in a gas - air
- Gas in a liquid - aerated drink
Matter
Matter is all around us. On the other hand, it can be defined as anything that has mass and occupies space. The air you are breathing is also a matter. The water we drink, the food we eat, the clothes we wear, and the air we breathe every day, each of these things has its own mass and volume.
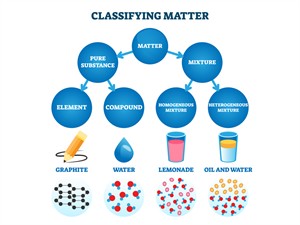
Classification of matters
Based on composition: Matter is classified as
- Pure substance
- Impure substance or mixture
i. Pure substance:
Pure substances are those that are completely made up of one form of particle. Physical processes cannot distinguish pure substances from other forms of matter.
The substances that are made up of only one kind of particle are known as pure substances. These are substances that cannot be separated into any other kinds of matter by any physical process.
Pure substances are further classified into elements and compounds.
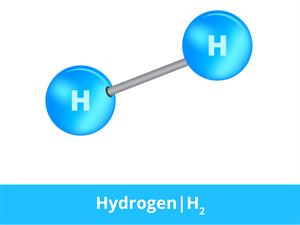
Hydrogen molecule
In hydrogen gas, the substance is made of the same atoms. Thus, two hydrogen atoms bond together to form a hydrogen molecule which is in a gaseous state.
Other examples of pure substances include sugar, salt, and diamond. Crystals, in general, are known as pure substances.


Pure gold (\(24\) carat)
ii. Impure substances:
In a fruit salad, \(4\) to \(5\) kinds of fruits are mixed together. It is an example of an impure substance, as we can separate it by the handpicking into separate fruits.

Fruit salad
A combination of more than one substance is known as an impure substance or a mixture. A physical process can separate mixtures.

Lemon juice with honey and water
In fresh juice, fruit is mixed with water and sugar. There are three different kinds of substances present in it, so it is called a mixture.
A substance formed by chemical combination is called a compound, while a substance formed by a physical combination is called a mixture. Physical methods can separate a mixture.
Example: Seawater, air, clay water, milk, and cereals.
Electrolysis of water:
When you observe a glass of water, it may appear to be simple and ordinary at first glance. However, this clear liquid actually hides a fascinating partnership between two distinct gases: hydrogen and oxygen. How do we prove this? One effective method is through the process of 'electrolysis of water', which separates water into its elemental components.
Let us engage in an activity to learn and understand the nature of water as a compound and its elemental components.
Activity: Electrolysis of water
The process in which water is decomposed by passing an electric current is called electrolysis of water. When an electric current is used for decomposing a substance, it is called electrolysis.
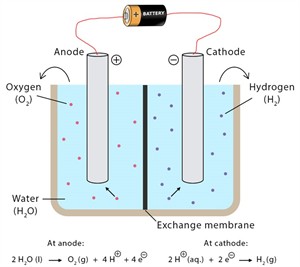
Electrolysis
Materials required:
-Plastic mug
-Rubber stopper
-Carbon electrodes (anode & cathode)
-Battery
-Water
-Dilute sulphuric acid and
-Test tube
Experimental procedure:
Step 1: Take the plastic mug and drill two holes at its base.
Step 2: Fit the two rubber stoppers in these holes.
Step 3: Insert the carbon electrodes (anode & cathode) into these rubber stoppers.
Step 4: Connect these carbon electrodes to a \(6\)-volt battery.
Step 5: Now, fill the plastic mug with water up to a level that allows the carbon electrodes to be immersed.
Step 6: Add a few drops of sulphuric acid to this water.
Step 7: Take two test tubes and fill them with water. Then, invert these test tubes over the carbon electrodes.
Step 8: Now, switch on the current from the \(6\)-volt battery.
Step 9: Leave the apparatus undisturbed for some time.
Observation:
- There will be the formation of bubbles at both electrodes in the test tubes (which means gas is forming or being liberated from the water)
- Due to the formation of bubbles, water begins to move in the test tube.
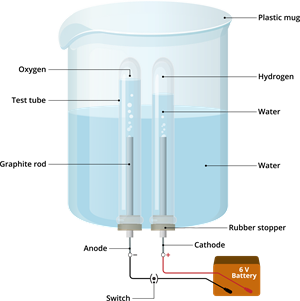
Electrolysis of water
Confirmation test of the liberated gases:
Once these test tubes are filled with the gases, carefully remove them from the mug. Place a glowing candle near the mouths of the test tubes to determine which gas is present in each.
Important!
The teacher must carefully carry out this step.
Result:
- When we bring a glowing candle close to the mouth of one of the test tubes, the gas in the test tube burns with a pop sound, indicating the presence of hydrogen.
- When we bring a burning candle closer to the mouth of another test tube, the candle burns brightly, indicating that the test tube contains oxygen.
- The gas collected at the anode (negative) is oxygen, and the gas collected at the cathode (positive) is hydrogen.
The volume of the gas obtained in both the test tubes is not the same. The volume of hydrogen gas is twice that of oxygen gas contained in the other test tube.
Decomposition reaction:
\(2H_2O\)(l) \(2H_2\)(g) + \(O_2\)(g)
(Water) (Hydrogen) + (Oxygen)
Conclusion:
In this reaction, water decomposes to form hydrogen and oxygen under suitable conditions. In this case, the electricity is a suitable condition for the water to decompose. In the compound water, the hydrogen and oxygen are combined in a \(2:1\) ratio.
Reference:
https://www.chemistrylearner.com/electrolysis-of-water.html