Have you noticed that sugar dissolves faster in hot milk than in cold milk? This happens due to changes in solubility and concentration.
In our daily life, we use many solutions like lemonade, salt water, and medicines. The amount of solute present in a solution is called its concentration, which tells how strong or dilute it is.
Solubility depends on factors such as temperature, and this relationship can be understood using solubility curves.
Let us learn how to express concentration and understand solubility and the effect of temperature on it.
The Concentration of a Solution:
The concentration of a solution is the amount of solute present in a given amount of solvent or solution. Thus, the proportions of solute and solvent present in a solution determine the concentration of the solution.
Dilute and concentrated solutions:
Depending on the amount of solute in the solution, it may be called a dilute or concentrated solution.
1. Dilute solution: A dilute solution contains a small amount of solute in a given amount of solvent .

Dilute solution
2. Concentrated solution: A concentrated solution contains a large amount of solute in a given amount of solvent .

Concentrated solution
The distinction between dilute and concentrated solutions is qualitative. It does not give the exact amount of solute present and is often observed through physical properties such as colour, density, etc.
To express concentration more accurately, quantitative methods are used.
Methods of Expressing Concentration:
The concentration of a solution can be expressed mathematically as:
The concentration of a solution can be expressed using the following methods:
| \(\text{Concentration of solution}\) \(=\) \(\frac{\text{Amount of solute}} {\text{Amount of solvent or solution}}\) |
The concentration of a solution can be expressed using the following methods:
- Mass by mass percentage
- Mass by volume percentage
- Volume by volume percentage
1. Mass by mass percentage.
We can find the mass percentage of a solution by dividing the mass of solute by the mass of the solution and multiply the product by \(100\).
| Mass by mass percentage \(=\) \(\frac{\text{Mass of solute}}{\text{Mass of solution}}\) \(\times100\) |
The mass percentage is commonly expressed as \(w/w\) (weight / weight)
Similarly, the concentration of solution in ointments, antacids, soaps, and other products is expressed as w/w.
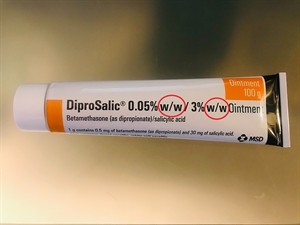
Ointment
2. Mass by volume percentage.
We can find the mass volume percentage of a solution by dividing the mass of solute by the volume of solution and multiply the product by \(100\).
| Mass by volume percentage \(=\) \(\frac{\text{Mass of solute}}{\text{Volume of solution}}\)\(\times100\) |
3. Volume by volume percentage.
We can find the volume percentage of a solution by dividing the volume of solute by the volume of solution and multiply the product by \(100\).
|
Volume by volume percentage \(=\) \(\frac{\text{Volume of solute}}{\text{Volume of solution}}\)\(\times100\)
|
Volume percentage is commonly expressed as \(v/v\) (volume / volume). In case if both the solute and the solvent are liquids, this method is used.
The concentration of the ingredients is expressed as \(v/v\) in commercial products that we come across in our daily lives, such as syrup solutions, mouthwash, antiseptic solutions, household disinfectants, and so on.
Solubility of a Substance:
Solubility is the maximum amount of a solute that can dissolve in a given amount of solvent at a specific temperature.
Types of Solutions Based on Solubility:
1. Unsaturated Solution:
An unsaturated solution is one that has not reached its maximum solubility. This means that more solute can still be dissolved in the solution at a given temperature.
2. Saturated solution:
A saturated solution is one in which no more solute can be dissolved at a given temperature.
3. Supersaturated solution:
A supersaturated solution is one that contains more solute than a saturated solution at a given temperature. Such solutions are usually unstable and may crystallize when disturbed.
Factors affecting solubility:
1. Nature of solute and solvent:
Solubility depends on the type of solute and solvent. Water can dissolve many substances, but not all. A solution in which water is the solvent is called an aqueous solution.
Scientists use the rule “like dissolves like”.
- Polar substances dissolve in polar solvents
- Non-polar substances dissolve in non-polar solvents
Examples:
- Common salt dissolves in water
- Salt does not dissolve in oil

Iodine in a) carbon tetrachloride b) water
Non-polar compounds, do not dissolve in polar solvents, and polar compounds do not dissolve in non-polar solvents.
2. Temperature:
(a) Solubility of Solids in Liquids:
- When temperature increases, solubility of most solids increases
- When temperature decreases, solubility decreases
This is because at higher temperature, particles move faster and dissolve more easily, while at lower temperature, particles move slowly and dissolve less.
Example:
Sugar dissolves faster in hot water than in cold water.
(b) Solubility of Gases in Liquids:
- When temperature increases, solubility of gases decreases
- When temperature decreases, solubility of gases increases.
This is because heating gives energy to gas particles, allowing them to escape from the liquid, while cooling keeps the gas particles dissolved in the liquid.
Examples:
- Bubbles form when water is heated
- Cold water has more dissolved oxygen, so fish survive better
3. Pressure (Only for Gases):
Increasing pressure leads to more gas dissolving, while decreasing pressure leads to less gas dissolving.
This is because high pressure forces gas particles to stay dissolved in the liquid.
Example:
Carbon dioxide is dissolved in soft drinks under high pressure.
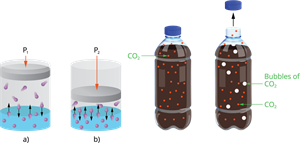
Effect of pressure on solubility
Solubility curve:
A solubility curve is a graph that shows how the solubility of a substance changes with temperature. It helps us understand how much solute can dissolve in water at different temperatures.
In a solubility curve:
- The \(X\)-axis represents temperature (\(°C\))
- The \(Y\)-axis represents solubility (\(g\) of solute in \(100\ g\) of water)
For most solid substances, solubility increases with increase in temperature, which means more solute can dissolve in hot water than in cold water.
Points on the solubility curve represent different types of solutions:
- A point on the curve represents a saturated solution (maximum solute dissolved).
- A point below the curve represents an unsaturated solution (more solute can still dissolve).
- A point above the curve represents a supersaturated solution (more solute than normal, unstable).

Solubility curve