Most substances around us are not pure and are found as mixtures. These mixtures can be of two types: homogeneous (same throughout) and heterogeneous (different parts can be seen).
Heterogeneous mixtures can be separated easily using simple methods like handpicking, sieving, and filtration, which we use in daily life. But homogeneous mixtures cannot be separated so easily. They require special separation techniques.
Principle of seperation techniques:
Separation techniques are based on differences in physical properties of the components of a mixture, These properties include differences in solubility, boiling point, density, particle size, magnetic nature, and adsorption behaviour. By making use of these differences, the components of a mixture can be separated without bringing about any chemical change in them.
Separating a pure solid from its solution is not always possible by simple methods like evaporation, as the solid obtained may contain impurities. To obtain pure crystals of a substance, a special technique called crystallisation is used.
Crystallisation:
A solution of copper sulphate looks uniform throughout. How can we separate and obtain its solid crystals again?
Principle:
Crystallisation is based on the difference in solubility at different temperatures.
Process:
When the hot concentrated liquid is cooled, it forms its crystal, and this process is known as crystallisation. In this process, all the soluble impurities are removed.
Crystallisation of Copper Sulphate:
- Take impure copper sulphate in a beaker.
- Add water and heat to dissolve it.
- Filter to remove insoluble impurities.
-
To get a saturated solution, evaporate the water from the copper sulphate solution.
-
Cover the solution with filter paper and set it aside to cool slowly at room temperature for a day.
- Blue crystals of copper sulphate are formed.


Copper sulphate solution Crystals of copper sulphate after crystallisation
Applications:
- Purification of salts from impure samples.
- Preparation of pure chemicals in laboratories.
- Helps in obtaining large, well-defined crystals for study and use
- Used in the production of alum and copper sulphate crystals
Separating a liquid from a solid-liquid mixture or separating liquids from a liquid-liquid mixture is not possible by simple methods like filtration. For this purpose, a special technique called distillation is used.
Distillation:
Sea water contains dissolved salts and looks uniform. How can we separate pure water from it?
Principle:
Distillation is based on the difference in boiling points of the components of a liquid mixture.
Process:
In distillation, the liquid mixture is heated so that the component with a lower boiling point vaporises first. The vapour is then cooled and condensed back into liquid form, which is collected separately. In this way, the components of the mixture are separated without any chemical change.
Types of distillation:
i. Simple Distillation: Simple distillation is used when the difference in boiling points of the liquids is large or when a liquid is to be separated from dissolved solids. In this method, the liquid is heated to form vapour, which is then cooled and collected as pure liquid, leaving behind the impurities.
Example: Separation of pure water from salt solution.
ii. Fractional distillation: Fractional distillation is used when two or more liquids have close boiling points. In this method, a fractionating column is used, which allows repeated condensation and vaporisation. This helps in separating the liquids more effectively based on their boiling points.
Example: Separation of different components of petroleum.
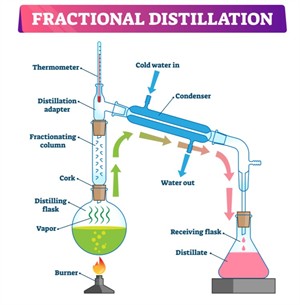
Fractional distillation
Applications:
- Used to convert seawater into drinking water.
- Used to refine crude oil into different fractions.
- Used to purify alcohol.
- Used to obtain distilled water for laboratories and hospitals.
- Used to separate liquids with different boiling points.
Separating the different coloured components of a mixture, such as dyes in ink, is not possible by simple methods like filtration or evaporation. For this purpose, a special technique called chromatography is used.
Chromatography:
Black ink appears to be a single colour. How can we separate it into its different coloured components?
Principle:
Chromatography is based on the difference in adsorption and solubility of the components of a mixture in a stationary phase and a mobile phase.
Process:
In chromatography, a small spot of the mixture is placed on a strip of paper (stationary phase). The lower end of the paper is dipped in a suitable solvent (mobile phase). As the solvent moves up the paper, it carries the components of the mixture along with it. Different components move at different speeds depending on their solubility and adsorption, and thus get separated at different positions on the paper.
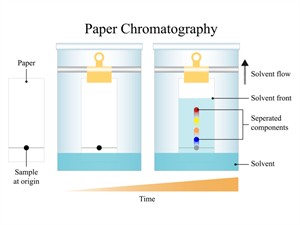
Paper chromatography
Applications:
- Colours in a dye can be separated using this method.
- Natural pigments can be separated using this method.
- Medicines derived from blood can be separated.
- Handle chemicals carefully and avoid direct contact.
- Use protective equipment like gloves and goggles when required.
- Be careful while heating substances during distillation or crystallisation.
- Do not inhale vapours or fumes directly.
- Handle glass apparatus like beakers and test tubes with care.
Chemistry in Daily Life and Society:
Separation techniques are widely used in our daily life and traditional practices. For example, salt is obtained from seawater by evaporation, and water is purified for drinking. Traditional distillation methods have been used in India for preparing herbal extracts and perfumes.
Indian scientists like Dilip Mahalanabis improved public health by promoting simple solutions like oral rehydration therapy, showing the importance of chemistry in everyday life.