Have you ever wondered what everything around us is made of, from the air we breathe to the phone in your hand? Whether it’s the food we eat or the clothes we wear, everything is made up of matter.
- Everything in the universe is made of matter.
- Atoms are the smallest particles of elements that cannot be broken down by chemically.
These ideas form the basis of all chemical studies and discoveries.
Historical background:
-
Around 500 BC, the Indian philosopher Maharishi Kanad proposed that if we go on dividing matter, we will ultimately get very small particles called Parmanu (atoms).
-
Around the same time, Greek philosophers Democritus and Leucippus suggested that matter is made of tiny indivisible particles called atoms (meaning “indivisible”).
Laws of chemical combination:
Chemistry deals with matter and the changes it undergoes, which follow specific rules called the laws of chemical combination, proposed by Antoine Lavoisier and Joseph Proust.
The laws of chemical combination are,
- Law of conservation of mass
- Law of constant proportions or law of definite proportions
Law of conservation of mass:
“The mass in an isolated system can neither be created nor destroyed but can be transformed from one form to another”
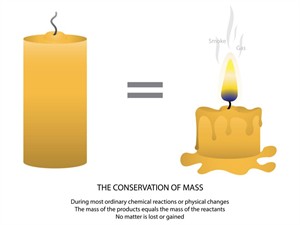
Law of conservation of mass
Law of constant (definite) proportion:
The elements are always present in definite proportions by mass in a chemical substance.
In water (\(H_2O\)), the ratio of the mass of hydrogen to oxygen is always \(1:8\), whether it is tap water, rainwater, or distilled water.
Postulates of Dalton's atomic theory:
-
All matter is made of tiny particles called atoms.
-
Atoms cannot be created or destroyed in chemical reactions.
-
Atoms of the same element are identical in mass and properties.
-
Atoms of different elements have different masses and properties.
-
Atoms combine in simple whole-number ratios to form compounds.
- The relative number and kinds of atoms are constant in a given compound.
Atoms:
The smallest unit of an element. Composed of subatomic particles:
- Protons (\(p^+\)) – Positive charge
- Neutrons (\(n^0\)) – No charge
- Electrons (\(e^−\)) – Negative charge
Nucleus contains protons and neutrons; electrons orbit in shells.
An atom is extremely small, with a size of about \(0.1\) \(nanometre\) (\(1 × 10^{-10}\) \(m\)).
Almost \(10\) \(million\) atoms lined up would make just \(1\) \(millimetre\).
Almost \(10\) \(million\) atoms lined up would make just \(1\) \(millimetre\).
Elements:
Substances that cannot be broken down into simpler substances by chemical methods.
Symbols of elements:
- John Dalton was the first scientist to use symbols to represent elements, but his symbols were pictorial, not letter-based.
- Jöns Jacob Berzelius, a Swedish chemist, later introduced the modern system of chemical symbols, using one or two letters derived from the element's English or Latin name.
We can see that each element has a name and a unique chemical symbol. Some elements' symbols are made up of the first letter of the name and a letter that appears later in the name.
Example:
i. Hydrogen - \(H\)
ii. Magnesium - \(Mg\)
Some elements have names derived from Greek or Latin, so their symbols may differ from their English names.
Example:
i. The symbol of iron is \(Fe\) from its Latin name Ferrum (\(Fe\)).
ii. Potassium symbol is \(K\) from Kalium (\(K\)).
iii. Sodium is \(Na\) from Natrium (\(Na\)).
Atomic mass:
The number of protons (\(p\)) in an atom is known as the atomic number (\(Z\)).
Mass number (A) = Number of protons + number of neutrons
According to the new IUPAC guidelines, the atomic mass unit is written as '\(u\)'.
Relative atomic mass:
In 1961, for a universally accepted atomic mass unit, carbon-\(12\) isotope was chosen as the standard reference for measuring atomic masses.
The relative atomic masses of all elements have been found with respect to an atom of carbon-\(12\). The relative atomic mass of the atom of an element is defined as the average mass of the atom, as compared to \(1/12\)th the mass of one carbon-\(12\) atom.
One atomic mass unit is a mass unit equal to exactly one-twelfth (\(1/12\)th) the mass of one atom of carbon - \(12\).
Molecules:
Atoms of most elements do not exist independently. Atoms form molecules and ions.
- A molecule can be defined as the smallest particle of an element or a compound that is capable of independent existence and shows all the properties of that substance.
- A molecule is made up of two or more atoms chemically bonded together. These atoms may be of the same element or of different elements.
Examples:
- Water (\(H_2O\)): \(2\) Hydrogen atom + \(1\) oxygen atom
- Oxygen (\(O_2\)): \(2\) oxygen atoms
Molecules of elements:
Molecules made of the same-type atoms are called molecules of elements.
| Type | Example | Symbol | Atomicity |
| Metals | Copper | \(Cu\) | Monoatomic |
| Sodium | \(Na\) | Monoatomic | |
| Non-metals | Hydrogen | \(H_2\) | Diatomic |
| Ozone | \(O_3\) | Triatomic | |
| Phosphorus | \(P_4\) | Tetra-atomic |
Atomicity: The number of atoms present in one molecule of an element.
Molecules of compounds:
When atoms of different elements combine chemically in a fixed ratio, they form molecules of compounds.
| Compound | Combining elements | Ratio by mass |
| Water (\(H_2O\)) | Hydrogen, Oxygen |
\(1:8\)
|
| Carbon dioxide (\(CO_2\)) | Carbon, Oxygen |
\(3:8\)
|
Ions:
When a neutral atom gains or loses electrons, it becomes an ion, which carry an electric charge.
- Cations: Positively charged ions formed by loss of electrons.
Example: \(Na\) → \(Na^+\) + \(e^−\)
- Anions: Negatively charged ions formed by the gain of electrons.
Example: \(Cl\) + \(e^−\) → \(Cl^-\)
The process of forming ions by gaining or losing electrons is called ionisation.
Sodium chloride (\(NaCl\)) – Formed from \(Na^+\) (cation) and \(Cl^−\) (anion).
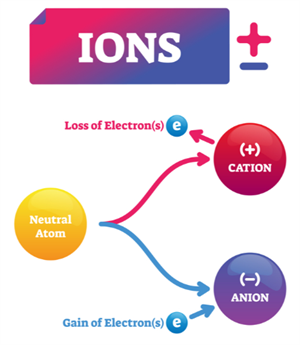
Ions
Chemical formulae:
A chemical formula is a symbolic representation of a compound showing the elements present and the number of atoms of each element. Before writing a chemical formula, it is important to understand the symbols and valency of elements.
Valency:
Valency is the combining capacity of an element. It tells us how many atoms of one element can combine with atoms of another to form a compound.
For example, hydrogen has a valency of \(1\), oxygen has \(2\), and nitrogen has \(3\).
Rules for writing a chemical formula:
-
Balance valencies or charges of combining elements or ions.
-
Write the symbol of the metal (positive ion) first, followed by the non-metal (negative ion).
-
Use brackets when more than one group of ions is needed.
Let us now see a few examples to understand how to write a chemical formula for a compound.
Formulae of simple compounds:
The constituent elements and their valencies are written down in the chemical formulae for compounds, as shown below.
The valencies of the combining atoms must then be crossed.
We know the symbol for hydrogen \(H\) and oxygen \(O\).
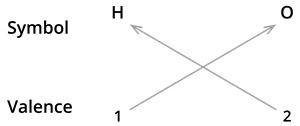
Formula: \(H_2O\)
Note: The ions' charges are also not specified in the chemical formula.
2. Formula of aluminium oxide:
We know the symbol for aluminium \(Al\) and oxygen \(O\).
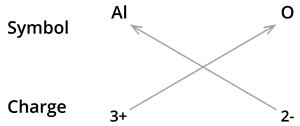
Formula: \(Al_2O_3\)
3. Formula of calcium oxide:
We know the symbol for Calcium \(Ca\) and oxygen \(O\).
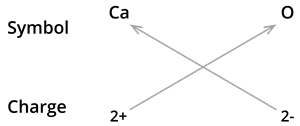
Formula: \(CaO\)
4. Formula of calcium hydroxide:
We know the symbol for calcium \(Ca\) and hydroxyl groups \(OH\).
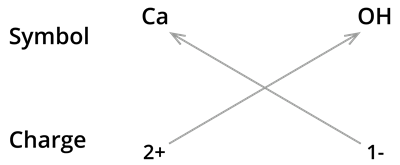
Formula:
Note:
-
The formula of calcium hydroxide is and not \(CaOH_2\). We use brackets when we have two or more of the same ions in the formula. Here, it indicates that there are two hydroxyl \((OH)\) groups joined to one calcium atom \(Ca\). And also, remember that brackets are not needed if only one ion is present.
Molecular mass:
The molecular mass of a substance is the sum of the atomic masses of all the atoms present in one molecule. It is expressed in atomic mass units (u).
Example: Molecular mass of water,
\(H_2O\) \(= (2 × 1) + (1 × 16)\)
\(= 18 u\)
Formula unit mass:
- For ionic compounds, we use the term formula unit mass instead of molecular mass.
- It is the sum of the atomic masses of all atoms in the formula of an ionic compound.
Example: Formula unit mass of sodium chloride,
\(NaCl\) \(= (1 × 23) + (1 × 35.5)\)
\(= 58.5 u\)
By understanding these concepts, we can explain how atoms form molecules and compounds, represent them with chemical formulae, and calculate their molecular or formula unit masses, building a strong foundation for exploring chemical reactions and equations.