Generally, we know that matter occurs in three states such as solid, liquid and gas. Let us now classify matter based on its composition.
Classification of matter:
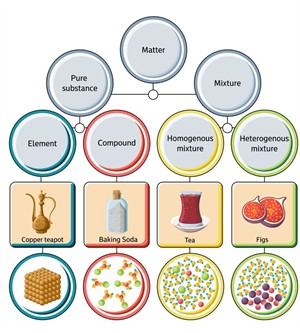
Classification of matter
Pure substance:
The substances which are made up of only one kind of particles are known as pure substances. These are substances that cannot be separated into any other kinds of matter by any physical process.
Example: Salt, sugar, and baking soda.
Based on the kind of atom, we classify pure substances as
- Elements
- Compounds
1. Elements:
Elements are the fundamental substances that cannot be separated into simpler substances by any chemical method.
i. Metals - Metals are elements that are generally hard, shiny, and good conductors of heat and electricity. They are malleable (can be hammered into thin sheets) and ductile (can be drawn into wires). Most metals have high melting and boiling points and show metallic lustre. Mercury is an exception among metals, as it exists as a liquid at room temperature.
Example: Gold, copper, silver, iron, aluminium, and zinc
ii. Non-metals - Non-metals are elements that are generally soft or brittle in the solid state and do not have metallic lustre. They are poor conductors of heat and electricity and usually have low melting and boiling points. Many non-metals exists as gases, while some are solids or liquids. Bromine is the only non-metal that exists as a liquid at room temperature. Diamond is an exception among non-metals because it is extremely hard and has a non-metallic lustre.
Example: Oxygen, nitrogen, sulphur, and diamond
iii. Metalloids - Substances that show the characteristics of both metals and non-metals are known as metalloids.
Example: Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), Tellurium (Te), and Polonium (Po)
2. Compounds:
It is a form of matter created by combining two or more elements in a specific mass ratio. To decompose it into its constituent components, we use chemical methods.
Example: Water (\(H_2O\)), nitrogen dioxide (\(NO_2\)), salt (\(NaCl\)), and so on.
Impure substances:
The physical combination of more than one kind of substance is known as an impure substance or mixture. Mixtures can be separated by physical processes.
Mixtures are classified into two types,
| Homogeneous mixture | Heterogeneous mixture |
| All of the mixture's components are blended evenly. |
All of the mixture's components aren't evenly distributed.
|
| There are no visible separate barriers. | There are noticeable separate lines. |
| There is just a single-phase solution. |
There are two or more phase in the solution.
|
|
Example: Salt dissolved in water
|
Example: Sand water, oil in water, chalk in water
|
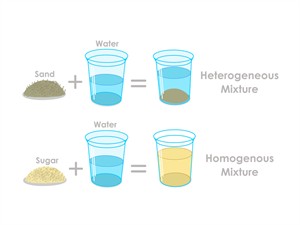
Types of mixtures
Difference between mixture and compound:
| Mixture | Compound |
| Constituents do not exist in a fixed ratio. | Elements are present in a fixed mass ratio. |
| Can be homogeneous or heterogeneous | Usually homogeneous |
| Constituents can be separated by physical methods | Constituents can only be separated by chemical methods |
| Constituents retain their identities and original properties | Constituents lose their individual identities and properties |
| Example: Iron + Sulphur | Example: Iron sulphide |
Solutions:
A solution is a mixture of two or more substances that appears to be uniform in appearance. A solution is a homogeneous mixture of two or more substances.
Types of solutions:
1. Solids in a liquid solution: Salt solution, sugar solution
Solute: Salt is the solute.
Solvent: Water is the solvent.
2. Gas in a liquid solution: Aerated drinks
Solute: Carbon dioxide (gas) is the solute.
Solvent: Water (liquid) is the solvent.
3. Solids in a solid solution: Alloys
Brass, for example, is made up of about \(30\)% zinc and \(70\)% copper.
4. Liquids in a liquid solution: Lemonade
Solute: Lemon extract is the solute.
Solvent: Water is the solvent.
5. Gas in a gas solution: Natural gas, air
Oxygen \(21\)% and nitrogen \(78\)% are the two primary constituents present in air along with small amounts of ither gases like argon and carbon dioxide.
The Concentration of a Solution:
A solution is a mixture in which one substance dissolves fully in the other. The solute is the material that dissolves. The solvent is the substance in which the solute dissolves.
a. Depending on the amount of solute in the solution, it may be called a dilute or concentrated solution.
1. Dilute solution: Dilute solution refers to a solution with a minimal volume of solute. This can also be known as an unsaturated solution.
2. Concentrated solution: A concentrated solution is a solution that contains a significant volume of solute.
b. Based on the dissolving limit of solvent and solute, the saturation can be classified into two types as a saturated solution and an unsaturated solution.
1. Saturated Solution: At a given temperature, a saturated solution is one in which no more solute can be dissolved.
2. Unsaturated solution: If the added solute completely dissolves, leaving no substances remaining at the bottom is called an unsaturated solution.
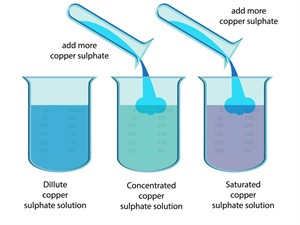
Concentration of solutions
The formula below is used to calculate the concentration of a solution.
There are various ways to express the concentration of a solution, but here, we will learn these three well-known methods:
| \(\text{Concentration of solution}\) \(=\) \(\frac{\text{Amount of solute}} {\text{Amount of solvent or solution}}\) |
There are various ways to express the concentration of a solution, but here, we will learn these three well-known methods:
- Mass by mass percentage
- Mass by volume percentage
- Volume by volume percentage
1. Mass by mass percentage:
We can find the mass percentage of a solution by dividing the mass of solute by the mass of the solution and multiply the product by \(100\).
| Mass by mass percentage \(=\) \(\frac{\text{Mass of solute}}{\text{Mass of solution}}\) \(\times100\) |
2. Mass by volume percentage:
We can find the mass volume percentage of a solution by dividing the mass of solute by the volume of solution and multiply the product by \(100\).
| Mass by volume percentage \(=\) \(\frac{\text{Mass of solute}}{\text{Volume of solution}}\)\(\times100\) |
3. Volume by volume percentage:
|
Volume by volume percentage \(=\) \(\frac{\text{Volume of solute}}{\text{Volume of solution}}\)\(\times100\)
|
We can find the volume percentage of a solution by dividing the volume of solute by the volume of solution and multiply the product by \(100\).
Properties of solution, colloid and suspension:
| Property | Solution | Colloid | Suspension |
| Nature of mixture | Homogeneous | Heterogeneous | Heterogeneous |
| Particle size | Less than \(1\) \(nm\) | Between \(1\) - \(100\) \(nm\) | Greater than \(100\) \(nm\) |
| Particle visibility | Not visible | Not visible | Visible to naked eye |
| Tyndall effect | No | Yes | Yes, but only until particles remain floating |
| Stability | Very stable | Stable | Unstable |
| Filtration | Cannot be seperated | Cannot be seperated by ordinary filtration | Can be seperated |
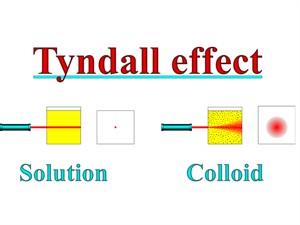
Tyndall effect on colloids
Types of colloidal solutions:
Classification of colloids based on the physical state of the dispersed phase and dispersion medium.
a. Dispersed phase: The dispersed phase is described as a phase that is scattered or present in the form of colloidal particles.
b. Dispersed medium: The dispersed phase is the medium in which colloidal particles are distributed.
|
S.No
|
Name
|
Dispersed phase
|
Dispersed medium
|
Example
|
|
1.
|
Solid sol
|
Solid
|
Solid
|
Colored glass, Gems, Alloys.
|
|
2.
|
Sol
|
Solid
|
Liquid
|
Paint, Fruit jellies, Dye, Ink, Egg white
|
|
3.
|
Aerosol
|
Solid
|
Gas
|
Smoke
|
|
4.
|
Gel
|
Liquid
|
Solid
|
Cheese, Butter.
|
|
5.
|
Emulsion
|
Liquid
|
Liquid
|
Milk, Oil in water, Mayonnaise, Face cream.
|
|
6.
|
Aerosol
|
Liquid
|
Gas
|
Fog, Mist, Clouds, Body sprays.
|
|
7.
|
Foam
|
Gas
|
Liquid
|
Soap lather, Shaving cream, Coffee froth
|
|
8.
|
Solid foam
|
Gas
|
Solid
|
Rubber, Sponge, Cake, Bread
|