Everything in this universe is made up of material, which scientists have termed "Matter".
Take a moment to look at our surroundings. We see a variety of things with different shapes, sizes and textures. The water we drink, the food we eat, the clothes we wear, and the air we breathe every day, each of these things have their own mass and volume.
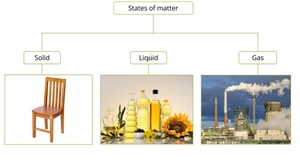
Different states of matter
But our modern-day scientists have proposed two types of classification of matter based on their physical properties and chemical nature.
Let us learn about matter based on its physical properties and chemical nature in details.
Matter's physical nature:
Matter contains "\(n\)" number of particles. The particles of matter are very small, which is beyond our imagination. The particles of matter has mass and occupies space.
Matter consists of atoms. Atom is the smallest particle that we cannot see through nacked eyes. An atom consists of particles of neutron, electron, and protons in which neutrons and protons are present in the nucleus of the atom and electrons are present outside of the nucleus.
Let see an example to understand this clearly.
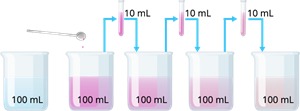
Potassium permanganate dissolution in water
The above experiment shows that just a few crystals of potassium permanganate can colour a huge volume of water. So we desired that there must be millions of very tiny particles in just one crystal of potassium permanganate, which keep on dividing themselves into smaller and smaller particles.
The particles of matter are very small they are very small beyond our thinking.
- Everything in this universe is made up of matter.
- Generally, matters occurs in solid, liquid and gas.
- Matter consists of tiny particles.
We can distinguish the characteristics of particles of matter into three types.
1. Particles of matter have space between them
Take a beaker with water and dissolve some salt with the help of a glass rod. Wait for a few seconds.
We already know that these particles are very tiny in nature. When we dissolve salt in water, the particles of salt get into the spaces between the particles of water. Thus, the water level is not raised.


Dissolving salt in water
2. Particles in the matter are continuously moving
To see this, drop a little blue ink into a bowl of water. Without stirring, the blue colour slowly spreads on its own. This shows that the particles of water and ink keep moving and mix by themselves.


Diffusion of ink
3. Particles of matter attract each other
The particles of matter attract each other due to the various forces acting between them. These forces keep the particles together. The strength of the forces of attraction varies from one kind of matter to another.


Attracting particles
States of matter
We can classify the matters into three types:
These are;
- Solid state
- Liquid state
- Gaseous state

States of matter
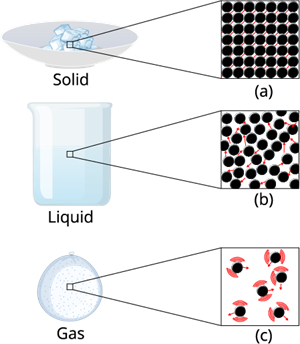
1. Solid state:
The objects that have a definite shape, distinct boundaries, and a fixed volume are called solid objects, or they are in a solid state.
The intermolecular force in the solid is the strongest force compared to the liquid and gas. This strongest intermolecular force makes the solid matter denser and gives it a certain shape and structure.

Rocks
2. Liquid state:
A liquid is an intermediate phase between a solid and a gas. It has a fixed volume but no fixed shape. A liquid can take the shape of any container it is kept in without changing its volume.

Fruit juices
The intermolecular force in a liquid is not as high as that of a solid, but it's higher than that of a gas.
3. Gaseous state:
A gas is a state of matter in which particles are very far apart and move freely in all directions. It has neither a fixed shape nor a fixed volume. A gas expands to fill the entire space of any container it is kept in.

Gas characteristics
Gases are highly compressible as compared to solids and liquids. In the gaseous state, the particles move about randomly at high speed.
Because of this property, we can use the gases in LPG in highly compressed condition. If we compress the gas particles, we can store it in a small container too.
We know that gas particles have more freely movable particles, so it can travel through the air easily. That's why the aroma of a food reaches to you even though you're in a separate room.
Intermolecular force on gases:
The intermolecular force in the gaseous substance is weaker compared to that in the liquid and solid. This weaker intermolecular force allows the gaseous matter to move freely in the air.
Intermolecular forces of matter