Have you ever wondered why ice turns into puddles of water on a sweltering day?
We already learn that matter occurs in three states - solid, liquid and gas.
Knowing that matters can change its states; when a state change occurs, the properties of substances also change. Similarly, if the state change is reversed, the substance will recover the properties which it had before.
Transformation of states:
1. Melting / Fusion: When the temperature increases, solid particles gain energy and break their fixed positions to form a liquid.
2. Vaporisation: The process by which a liquid converts its physical state to a gaseous state is known as vaporisation.
3. Condensation: The process of a gas converting its physical state to a liquid state is known as condensation. This is also called reverse vaporisation.
4. Freezing/Solidification: On cooling, liquid particles lose energy, move slowly, and get tightly packed to form a solid.
5. Sublimation: Some solids directly change into a gas on heating because their particles get enough energy to overcome attraction.
6. Deposition: When gas cools rapidly, its particles lose energy and directly form a solid without becoming liquid.

Transformations of states
Boiling point:
The temperature at which a liquid begins to boil at atmospheric pressure is known as its boiling point.
Melting Point:
The temperature at which a solid substance melts to become a liquid at atmospheric pressure is called the melting point of that substance.
Celsius and Kelvin:
The SI unit of temperature is Kelvin (K). Celsius (°C) is not an SI unit, but it is commonly used in daily life.
We use the symbol \(°C\) to represent the Celsius; similarly, we use \(K\) for Kelvin.
When the Celsius is \(0°\), the Kelvin will be \(273.15K\).
That is, .
Celsius to Kelvin:
To convert the temperature on the Celsius scale to the Kelvin scale, we have to add 273.15 to the given temperature.
Kelvin to Celsius:
To convert the temperature from the Kelvin scale to the Celsius scale, subtract \(273.15\) from the given temperature.
Absolute Zero:
The temperature at which there is no motion and no heat remains in the substance, and all the particles will freeze their motion.
Absolute zero is the temperature at which a thermodynamic system has the lowest energy. It corresponds to \(-273.15\) °C (Celsius)or \(-459.67\ \)°F (Fahrenheit).
Effects of pressure on states:
Solids have particles that are tightly packed, so applying pressure does not change their shape or volume.
Liquids have particles that are close but can move slightly, so pressure changes their volume very little.
Gases have particles that are far apart and move freely, so they can be easily compressed or expanded by changing the pressure.
Therefore, solids are almost incompressible, liquids are slightly compressible, and gases are highly compressible.
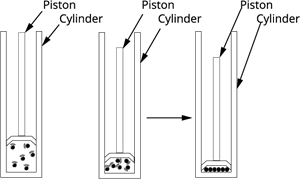
Effects of pressure
Evaporation:
Evaporation is the process by which molecules at the surface of a liquid gain enough energy to change into the gaseous state, below the boiling point.
Evaporation is a phenomenon acting on the surface. The evaporation process increases with the following:
1. An increase in surface area:
If the surface area of the liquid layer is increased, the rate of evaporation increases.
2. An increase in temperature:
When the temperature of the liquid increases, the particles in the liquid get enough kinetic energy to reach the liquid to vapour state.
3. A decrease in humidity (the amount of water vapour present in air):
When the amount of water in the air is significantly high, it decreases evaporation.
4. An increase in wind speed:
We might have seen that when the wind speed is high, the wet clothes will dry faster. As the wind speed increases, the particles of water vapour move away with the wind, decreasing the amount of water vapour in the surroundings.

Evaporation