Atoms make up everything around us, from the air we breathe to our daily objects. Let’s take a moment to recall what we already know about atoms before exploring their structure in greater detail.
- An atom is the smallest particle.
- Everything around us is made of atoms.
- Atoms are indivisible.
According to Dalton, atoms are indivisible. However, later experiments proved that atoms contain smaller particles called subatomic particles such as electrons, protons, and neutrons.
|
Subatomic particles
|
Scientists name
|
Symbol
|
Charge
|
Mass (amu) |
|
Electrons (cathode rays)
|
J. J. Thomson
|
\(e^-\)
|
\(−1\)
|
\(0\)
|
|
Protons (canal rays)
|
Eugen Goldstein
|
\(p^+\)
|
\(+1\)
|
\(1\)
|
|
Neutron
|
James Chadwick
|
\(n^0\)
|
No charge
|
\(1\)
|
To explain how these particles are arranged inside an atom, J.J. Thomson gave the first model of the atom.
Thomson's model of atom:
- The electrons are embedded in a positively charged sphere that represents an atom.
- The magnitudes of the negative and positive charges are the same. Hence, the atom is electrically neutral as a whole.
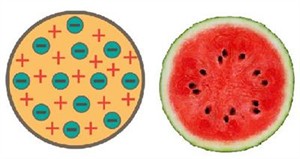
Thomson's atom (Watermelon) model
Limitations of J.J. Thomson’s model of the atom:
- It could not explain how positive charge and electrons were arranged inside the atom. It also failed to explain the stability of an atom.
- There was no mention of an atom's nucleus in the theory.
- It was unable to explain rutherford's scattering experiment.
Rutherford model:
Ernest Rutherford performed the alpha particle (\(He^{2+}) scattering experiment using a thin gold foil.
Observations:
- Since most alpha particles passed through the gold foil without being deflected, most of the space inside the atom is empty.
- Only a few particles were deflected from their direction, suggesting that the atom's positive charge uses very little space.
- Just a small percentage of alpha particles were deflected by \(180°\), showing that the gold atom's positive charge and mass were concentrated in a very small volume within it.
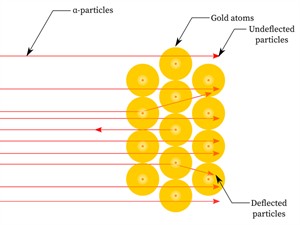
Scattering of α-particles by a gold foil
Features of the atom:
- The positive center of the atom is known as the nucleus.
- All the mass is concentrated on the nucleus, around which the electrons circulate in the well-defined orbit, much like planets revolving around the Sun.
- The size of the nucleus is less than the atom.
Drawbacks of Rutherford's atomic model:
As electrons revolve in orbit, they accelerate and lose energy. Thereafter, they fall into the nucleus. If this happened, the atom would collapse and matter would no longer exist. Thus, Rutherford's model failed to explain the stability of the atom.
To overcome Rutherford’s drawbacks, Niels Bohr proposed a new model.
Postulate of Niel's Bohr's model:
- The electrons revolve around the nucleus in a specific orbit, and these orbits are associated with definite energies called shells or energy levels.
- The electrons do not emit energy when revolving in specific orbits.
These shells, or energy levels, or orbits, are represented by the letters \(K\), \(L\), \(M\), \(N\) or by the numbers \(1\), \(2\), \(3\), \(4\).
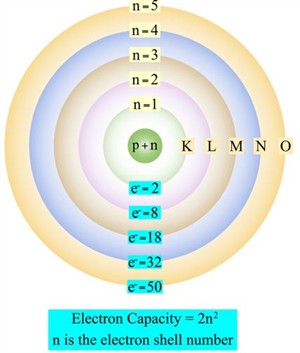
Bohr's atom model (electron shell diagram)
Distribution of electrons in orbits or shells:
| Energy levels | Shells | Maximum electrons | Electron capacity |
|
\(1\)
|
\(K\)
|
\(2\) × \((1)^2\) = \(2\)
|
\(2\)
|
|
\(2\)
|
\(L\)
|
\(2\) × \((2)^2\) = \(8\)
|
\(8\)
|
|
\(3\)
|
\(M\)
|
\(2\) × \((3)^2\) = \(18\)
|
\(18\)
|
|
\(4\)
|
\(N\)
|
\(2\) × \((4)^2\) = \(32\)
|
\(32\)
|
Electrons fill the shells gradually in the order of increasing energy. Unless the inner shells are completely filled, electrons cannot enter the next shell. The order of filling is: \(K\) < \(L\) < \(M\) < \(N\).
Valence electrons:
The electrons that are found in an atom's outermost orbit are called valence electrons. These electrons determine the chemical reactivity and bonding behavior of an element.
The charge of an atom is decided by the loss or gain of electrons. An atom becomes positive when it loses an electron and negative when it gains an electron.
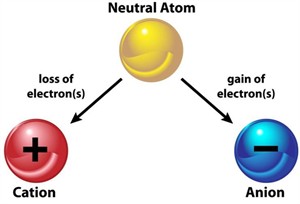
Formation of anion and cation from a neutral atom
Stable state:
Atoms try to achieve a stable electronic configuration.
-
If an atom has one shell, it is stable with \(2\) electrons (duplet rule).
-
If an atom has more than one shell, it is stable with \(8\) electrons in the outermost shell (octet rule).
Noble gases like \(He\), \(Ne\) and \(Ar\) are already stable.
Valency:
Valency is the number of electrons an atom can gain, lose, or share to achieve a stable electronic configuration.
Examples:
-
Oxygen (\(2,6\)) → gains \(2\) electrons → valency = \(2\)
-
Magnesium (\(2,8,2\)) → loses \(2\) electrons → valency = \(2\)
Formation of sodium chloride (\(NaCl\))
-
Sodium (\(2,8,1\)) loses one electron → \(Na^+\) (\(2,8\))
-
Chlorine (\(2,8,7\)) gains one electron → \(Cl^-\) (\(2,8,8\))
-
The electrostatic attraction between \(Na^+\) and \(Cl^-\) forms an ionic bond, resulting in sodium chloride (\(NaCl\)).
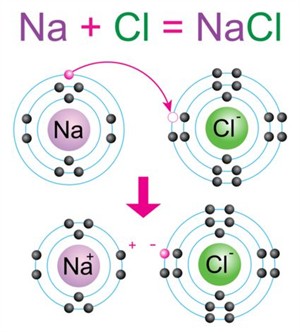
Formation of sodium chloride molecule
Atomic number:
The number of protons in the nucleus of an atom is known as its atomic number. The symbol "\(Z\)" stands for the atomic number. The atom of a different element has a different number of protons.
In a neutral atom, the number of protons is equal to the number of electrons.
Mass number:
The mass number or atomic mass of an atom is equal to the sum of the number of protons and neutrons present in the nucleus. It is represented by the symbol "\(A\)"
By rearranging the atomic mass formula, we can calculate the number of neutrons.
Isotopes:
We observed that some elements have the same atomic numbers but different mass numbers. These are known as isotopes. Their chemical properties are the same, but their physical properties are different.
Example:
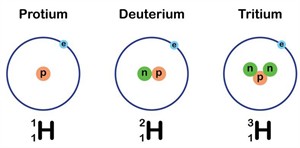
A hydrogen atom with the same atomic number but a different mass number
Application of Isotopes:
- The age of fossils, fuels, and dead organisms is determined using carbon-\(14\) isotopes.
- Cobalt-\(60\) is a radioactive isotope. It decays by emitting gamma rays, which are used to destroy cancer cells.
- In the treatment of goitre, an isotope of iodine is used.
- Blood flow is traced with sodium-\(24\) to identify whether there is an obstruction.
- Uranium-\(235\) is used as fuel in nuclear reactors.
Isobars:
Isobars are atoms with different atomic numbers but the same mass number. In other words, the nucleon count is the same, but the number of protons is different. Since they are different elements, their chemical properties are different.

Example for isobars
Isotones:
Isotones are atoms of different elements with different atomic numbers and mass numbers but the same number of neutrons.
Example: and
Number of neutrons in boron = \(11\) − \(5\) = \(6\)
Number of neutrons in carbon = \(12\) − \(6\) = \(6\)