Extraction of aluminium:
- Ore: Bauxite (\(Al_2O_3.2H_2O\))
- Nature of metal: Highly reactive, occurs in combined state
- Extraction: Aluminium is extracted from bauxite in two steps.
- Step I: Baeyer's process - Conversion of bauxite into alumina (\(Al_2O_3\))
- Step II: Hall's process - Electrolytic reduction of alumina (\(Al_2O_3\))
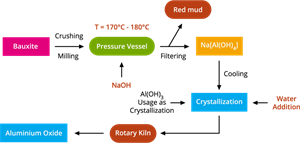
The flowchart of bayer's process
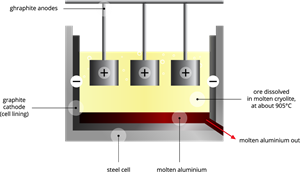
Pure aluminium extraction (Hall-Héroult) process
Extraction of Copper
- Ore: Copper pyrites (\(CuFeS_2\))
- Extraction: Copper is extracted from copper pyrites by the following steps, as shown in the diagram below.
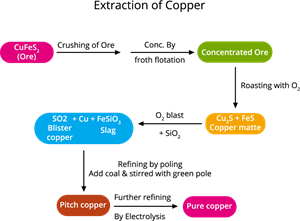
Copper extraction flowchart
Extraction of Iron
- Ore: Haematite (\(Fe_2O_3\))
- Extraction: Iron is extracted from haematite in the follwing steps.
- Step I: Concentration
- Step II: Roasting and calcination
- Step III: Smelting
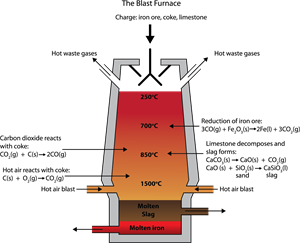
Blast furnace equipment
Chemical properties of aluminium, copper, and iron:
| Chemical property | Aluminium (\(Al\)) | Copper (\(Cu\)) | Iron (\(Fe\)) |
| Reaction with air |
\(4Al\) + \(3O_2\) → \(2Al_2O_3\)
\(2Al\) + \(N_2\) → \(2AlN\)
|
\(2Cu\) + \(O_2\) → \(2CuO\) | \(3Fe\) + \(2O_2\) → \(Fe_3O_4\) |
| Reaction with steam | \(2Al\) + \(3H_2O\) → \(Al_2O_3\) +\(3H_2\) | - | \(4Fe\) + \(4H_2O\) → \(Fe_3O_4\) +\(4H_2\) |
| Reaction with acids |
\(2Al\) + \(6HCl\) → \(2AlCl_3\) + \(3H_2\)
\(2Al\) + \(Con. 6H_2SO_4\) → \(Al_2(SO_4)_3\) + \(6H_2O\) + \(3SO_2\)
|
\(2Cu\) + \(4HCl\) + \(O_2\) → \(2CuCl_2\) + \(2H_2O\)
\(Cu\) + \(2H_2SO_4\) → \(CuSO_4\) + \(SO_2\) + \(2H_2O\)
|
\(Fe\) + \(2HCl\) → \(FeCl_2\) + \(H_2\)
\(4Fe\) + \(Con. 6H_2SO_4\) → \(Fe_2(SO_4)_3\) + \(3SO_2\) + \(6H_2O\)
|
| Reaction with alkali | \(2Al\) + \(2NaOH\) + \(2H_2O\) → \(2NaAlO_2\) + \(3H_2\) | - | - |
| Reaction with chlorine | \(2Al\) + \(3Cl_2\) → \(2AlCl_3\) | \(Cu\) + \(Cl_2\) → \(CuCl_2\) | \(2Fe\) + \(3Cl_2\) → \(2FeCl_3\) |
| Reducing agent | \(Fe_2O_3\) + \(2Al\) → \(2Fe\) + \(Al_2O_3\) + Heat | - | - |
| Corrosion behaviour | Protected by oxide layer | \(2Cu\) + \(O_2\) + \(CO_2\) + \(H_2O\) → \(CuCO_3.Cu(OH)_2\) | \(4Fe\) + \(3O_2\) + \(xH_2O\) → \(2Fe_2O_3.xH_2O\) |
Note: Dilute or concentrated nitric acid does not react with aluminium and iron, but renders them passive by forming a thin oxide film on their surface.
PYQ - Metal uses and acids
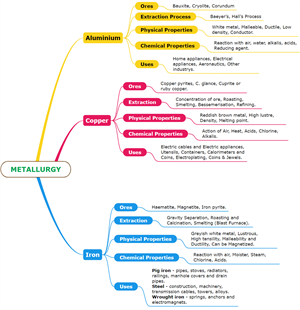
Properties and uses of \(Al\), \(Cu\) and \(Fe\)
Alloys:
An alloy is a homogeneous mixture of two or more metals or of one or more metals with certain non-metallic elements.
Amalgam:
An amalgam is a mixture of mercury and another metal. These alloys are formed by metallic bonding, which uses the electrostatic force of attraction between electrons and positively charged metal ions.
PYQ - Alloys
Types of alloys:
i. Ferrous alloy (Contains iron as main metal)
| Alloy | Composition | Uses |
| Stainless steel | \(Fe\) + \(C\) +\(Cr\) + \(Ni\) | Utensils, cutlery, automobile parts |
| Nickel steel | \(Fe\) + \(C\) + \(Ni\) | Cables, aircraft parts, propeller |
ii. Non-ferrous alloy (Do not contain iron as main material)
| Alloy | Composition | Uses |
| Brass | \(Cu\) + \(Zn\) | Electrical fittings, medals, decorative items, hardware |
| Bronze | \(Cu\) + \(Sn\) | Statues, coins, bells, gongs |
| Duralumin | \(Al\) + \(Mg\) + \(Mn\) + \(Cu\) | Aircrafts, tools, pressure cookers |
| Magnalium | \(Al\) +\(Mg\) | Aircraft, scientific instruments |
Corrosion:
It is a gradual destruction of metals by chemical or electrochemical reactions to the environment. It is a natural process that converts metal into its oxide, hydroxide or sulphide to lose its metallic characteristics.
Rusting:
Rust is chemically known as hydrated ferric oxide (it is formulated as \(Fe_2O_3. xH_2O\)). Rusting results in the formation of scaling reddish-brown hydrated ferric oxide on the surface of iron and iron-containing materials.
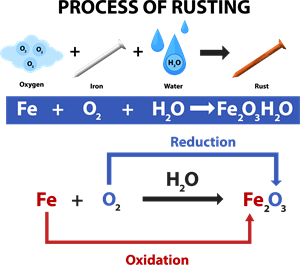
Rusting of iron
PYQ - Rust
Types of corrosion:
- Dry corrosion
- Wet corrosion
Methods of preventing corrosion:
- Alloying
- Galvanisation
- Electroplating
- Anodising
- Cathodic protection