Importance:
The chapter "Periodic Classification of Elements" is a core topic in chemistry and carries a weightage of \(4\) to \(6\) marks.
It helps students understand the systematic arrangement of elements based on,
- Atomic number and recurring properties,
- Explains trends in reactivity,
- Metallic character, and valency,
- Introduces the basics of metallurgy, including extraction, uses, alloys, and corrosion of metals.
Question distribution:
-
Part II (\(2\) mark) – One question
-
Part III (\(4\) mark) – One question
(Note: The exact mark distribution may vary slightly across examinations.)
Learning outcomes:
- Modern periodic law: Understand the classification of elements based on atomic number and periodic properties.
- Periodic table structure: Learn about periods, groups and the systematic arrangement of elements in the modern periodic table.
- Periodic trends: Understand how atomic radius, ionization energy, and electron affinity vary across a period and down a group in the periodic table.
- Metallurgy of metals: Learn about the occurrence, ores, extraction processes, physical properties, chemical properties and uses of aluminium, copper and iron.
- Alloys and corrosion: Understand the formation of alloys, their uses, corrosion, rusting and methods of prevention.
Introduction to elements:
Everything in the universe is made of matter. Different substances are formed by the combination of elements, each having unique properties. Scientists have classified elements based on their properties for easier study.
Modern Periodic Law
In \(1912\), Henry Moseley, showed that elements should be arranged by atomic number. The modern periodic law states that,
“The physical and chemical properties of elements are periodic functions of their atomic numbers.”
This law resolved inconsistencies present in Mendeleev’s periodic table.
In the modern periodic table, the horizontal rows are called periods, and the vertical columns are called groups. The modern periodic table consists of \(7\) periods and \(18\) groups.
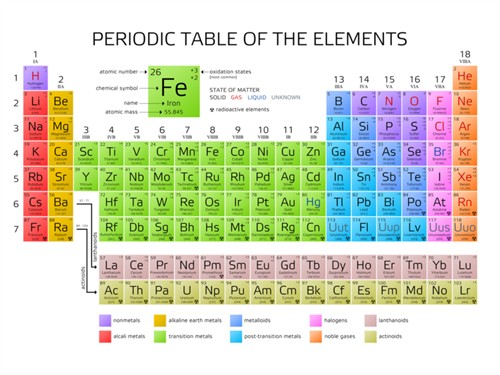
Periodic table of the elements
|
Group Number
|
Family
|
|
1
|
Alkali Metals |
|
2
|
Alkaline earth metals |
|
3 to 12
|
Transition metals |
|
13
|
Boron Family |
|
14
|
Carbon Family |
|
15
|
Nitrogen Family |
|
16
|
Oxygen Family (or) Chalcogen family |
|
17
|
Halogens |
|
18
|
Noble gases |
Periodic trends in properties
Periodicity:
The electronic configurations of elements help us to describe the periodic recurrence of physical and chemical properties. Anything which repeats itself after a fixed interval is called periodic, and this behaviour is called periodicity. Some of the atomic properties of the elements are periodic.
Periodic properties:
|
Periodic Property
|
In Periods
|
In Groups
|
| Atomic radius | Decreases | Increases |
| Ionic radius | Decreases | Increases |
| Ionisation energy | Increases | Decreases |
| Electron affinity | Increases | Decreases |
| Electronegativity | Increases |
Decreases
|
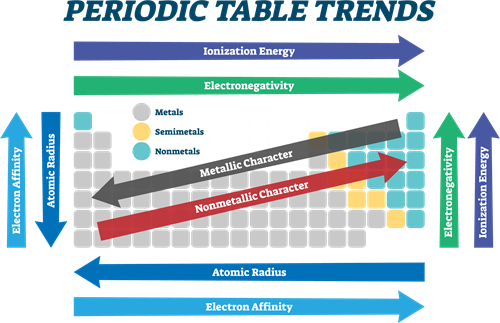
Periodic properties
Introduction to metallurgy
Metallurgy is a science of extracting metals from their ores and modifying the metals into alloys for various uses, based on their physical and chemical properties and their structural arrangement of atoms.
A metallurgical process consists of three significant steps, which are as follows:
i. Concentration or Separation of the ore
ii. Production of the metal
iii. Refining of the metal
Terminology in metallurgy:
- Mineral: A single compound or a complex mixture of different metal combinations found in the earth.
- Ore: A mineral that can be readily and economically extracted on a large scale.
- Mining: The process of extracting ores from the earth’s crust.
- Gangue/ Matrix: The rocky impurity associated with an ore.
- Flux: A substance added to the ore to decrease the fusion temperature and remove impurities.
- Slag: Fusible product formed when flux combines with gangue during metal extraction. (Flux + Gangue → Slag)
- Smelting: The process of reducing the roasted metallic oxide from the metal in its molten condition.
Types of separation or concentration of an ore:
- Hydraulic (gravity separation): Uses the difference in density between ore and gangue.
- Magnetic separation: Uses magnetic properties; ore or gangue is attracted by a magnet.
- Froth flotation: Metallic sulphide particles are wetted by oil; gangueis wetted by water.
- Chemical process (Leaching): Ore is dissolved using a reagent, impurities filtered out, then ore is precipitated from the solution.
The extraction of metal from metal oxide can be categorised into three types:
|
More reactive metals
|
Medium reactive metals
|
Less reactive metals
|
|
\(Na\), \(K\), \(Ca\), \(Mg\), \(Al\)
|
\(Zn\), \(Fe\), \(Pb\), \(Cu\)
|
\(Ag\), \(Hg\)
|
|
Electrolytic reduction of
metal oxide into metal |
Chemical reduction of metal
oxide into metal using coke |
Thermal decomposition of
metal oxide into metal |
Physical properties of metal:
| Property | Metals |
| Physical state |
Mostly solid at room temperature
Exception: Hg, Cs, Ga - Liquid near room temperature
|
| Lustre | Lustrous |
| Hardness |
Mostly hard
Exception: Na, K, Li - Soft
|
| Density |
High
Exception: Alkali metals (Li, Na, K)
|
| Melting and boiling point |
High
Exception: Hg, Cs, Ga, Na, K, Li
|
| Malleability and ductility |
Malleable and ductile
Exception: Hg and Zn
|
| Conductivity |
Good conductor of heat and electricity
Silver - Best conductor
Exception: Lead and mercury (Heat)
|
| Solubility |
Metals do not usually dissolve in liquid solvents.
|
Chemical properties of metal:
i. Valence electrons: Metal's atoms normally have one, two, or three electrons in their outermost (valence) shell.
ii. Formation of ions: Metals are electropositive as they lose electrons to produce positive ions.
iii. Discharge of ions: Metals are releases ions at the cathode during the electrolysis of their compounds.
iv. Atomicity: Metal molecules in the vapour state are generally monoatomic.
v. Nature of oxides: Metal oxides are usually basic (alkaline) in nature.