Shaanu and Gokul went to a restaurant with their friends and had delicious food. But, while eating, accidently, a bit of turmeric stain occurred in Shaanu's dress.
On the next day, she saw the same cloth when mixed with soap solution during washing; the stain turns into reddish-brown in colour. Do you know why?
When a cloth is washed with a soap (basic substance), the stain containing acid changes its colour to reddish-brown due to a neutralisation reaction. So when washed with excess water, the reddish-brown colour changes back to yellow.
When a cloth is washed with a soap (basic substance), the stain containing acid changes its colour to reddish-brown due to a neutralisation reaction. So when washed with excess water, the reddish-brown colour changes back to yellow.
Turmeric as an indicator:
Can you believe that there is an ingredient straight out of the kitchen that acts as a detective to investigate the nature of the solutions?
Yes, we have an agent yellow from our spices that's Turmeric!
Ready to witness the role of turmeric as an acid-base indicator!
Activity 1 - Let's create a unique card together to surprise your dad for Father’s Day
Materials Required:
-
Turmeric
-
Water
-
White paper and paintbrush
-
Soap solution
-
Lemon juice
Instructions:
Step 1: Mix the turmeric powder with water, and your golden yellow paint is ready.

Turmeric paste
Step 2: Apply turmeric paste on white papers and let it dry.

Paper painted with turmeric paste
Step 3: With the help of a paint brush or buds, write or paint your thoughts to make a card with lemon juice and soap solution on each paper, and watch what happens.
Observation:
What is the colour of the turmeric paste?
Ans: Yellow
Did any colour changes occur when painted with lemon juice?
Ans: No
Did any colour changes occur when painted with soap solution?
Ans: Turns reddish-brown
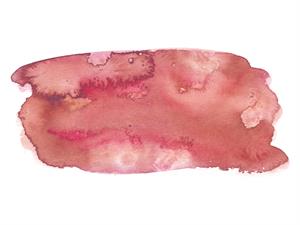
Painted over turmeric with soap solution
Can you name a solution which, when used, might change colour?
Ans: Baking soda solution
Conclusion:
Turmeric has a compound named curcumin, which is responsible for the colour change.
-
Acidic solution - Stays yellow
-
Basic solution - turns reddish-brown
- Neutral solution - Stays yellow
Interesting fact!
So far, we have been learning the indicator which allows us to know about the solution with the help of colour change. There is one more interesting factor, which indicates through smell, which is an olfactory indicator.
Olfactory Indicator:
The olfactory indicators are the substances which indicate a change in odour in acidic and basic media.
Example:
Onion, clove oil, garlic extract, and vanilla essence
When these substances are brought into contact with acids and bases, they convey the nature of the chemical, either becoming odourless or retaining the odour.
Activity: Testing solutions with olfactory indicator
Materials required:
- Onion, garlic
- Strips of clean cotton cloth
- Air-tight container
- Samples of acids and bases
Instructions:
Step 1: Take some garlic, finely chop it and add it to an air-tight container
Step 2: Add clean strips of cotton cloth to the same container, close and leave it overnight
Step 3: The next morning, remove the strips, and the strip smells like garlic
Step 4: Now test the strips with acid and base samples. To one strip, add a few drops of lemon juice, and to the other strip, add a few drops of soap solution. And check the odour of the strips. Perform the same for onoin
Observation:
In the acidic solution, the garlic-infused cloth strips retain the odour, while in the basic solution, they become odourless. The observations are listed as follows,
|
Smell in acid
|
|---|
|
Retains odour
|
|
Retains odour
|
|
Retains odour
|
|
Retains odour
|