The usage of iron and steel articles has become an integral part of our daily lives. People use iron for a wide range of everyday items, including bridges, ships, cars, truck bodies, window grilles, and many other articles.
Chemical reactions on the surfaces of shiny metals and other items cause them to lose their shine. When iron is exposed to wet air for an extended period, it corrodes, forming a coating called rust, a brown, flaky substance.
Activity: Testing conditions in which iron objects develop rust
Take a few shiny iron nails and place them in three different clean test tubes. Label them as test tubes 'A', 'B', and 'C'.
- In test tube \('A'\), pour enough water to partially immerse the iron nail, leaving some air space at the top.
- In test tube \('B'\), pour enough clean water (without dissolved air) to completely immerse the iron nail. Then, add a layer of oil on top of the water to cover it. Boiling the water removes dissolved oxygen, and the oil layer prevents atmospheric oxygen from dissolving back into the water.
- In test tube \('C'\), add some calcium chloride (a drying agent) and seal it tightly with a lid.
Place all three test tubes in a stable environment at room temperature, undisturbed.
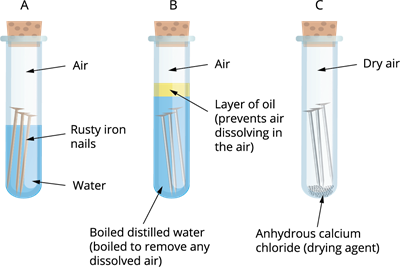
Testing of iron nails in different conditions
Observation:
Iron nails rust in test tube \('A'\), but not in test tubes 'B' or 'C', as you will observe. The nails in test tube 'A' are exposed to both air and water. The nails in test tube 'B' contains water without dissolved air, so ovygen is absent. While those in test tube 'C' are only exposed to dry air. Hence, rusting of iron requires both air and moisture.
Iron material loses its shine and turns into a red-brown, flaky substance when exposed to air and moisture for an extended period, a process known as rusting.
The chemical process of rusting is as follows,
\(Iron\) \(+\) \(Oxygen\) \(+\) \(Water\) \(\to\) \(Iron\) \(oxide\) (\(rust\))
\(4Fe + 3O_2 + xH_2O \to 2Fe_2O_3.xH_2O\)
Rusting occurs more rapidly in environments with high humidity (i.e., a high moisture content in the atmosphere) and a high salt content in the water.
Prevention of corrosion:
- Painting, oiling, greasing, and chrome plating, are all effective methods for preventing iron from rusting.
- Galvanisation is a process that coats steel and iron with a thin layer of zinc to prevent rusting. Even if the zinc coating on the galvanised item is damaged, it remains protected against rust.
- Alloying is an excellent way to improve a metal's properties. This method can be used to obtain the desired properties. When iron is combined with nickel and chromium, stainless steel is formed, a hard, rust-resistant metal.
Some other metals also get corroded in this way. Have you ever observed the colour of the coating formed on copper and silver metals?
i. When exposed to oxygen, carbon dioxide, and water vapour in moist air, copper undergoes corrosion and forms a greenish layer called patina (basic copper carbonate). The green coating is primarily due to the reaction of copper with carbon dioxide (\(CO_2\)) and moisture (\(H_2O\)) in the air, forming basic copper carbonate.
ii. Silver metal reacts with hydrogen sulphide gas present in the air to form silver sulphide. As a result, silver objects appear dull and black due to the layer of silver sulphide that is created on their surface.
\(Silver + Hydrogen\ sulphide \to Silver\ sulphide\)
i. Reaction of metals with water:
The reactivity of metals towards water differs. It is based on the reactivity of the metals,
- Sodium and potassium react vigorously with cold water, which liberates a lot of heat during the reaction. The reaction is exothermic.
Sodium + Cold water \(\to\) Sodium hydroxide + Hydrogen gas + Heat
- Calcium reacts with normal water to form calcium hydroxide and hydrogen gas. Magnesium reacts with hot water to form magnesium hydroxide and hydrogen gas.
Calcium + Normal water \(\to\) Calcium hydroxide + Hydrogen gas
- Iron, aluminium, and zinc do not react with normal, hot or cold water, but when they come into contact with steam, they produce metal oxide and hydrogen.
Iron + Steam (water vapour) \(\to\) Iron oxide + Hydrogen gas
Aluminium + Steam (water vapour) \(\to\) Aluminium oxide + Hydrogen gas
- Lead, copper, silver and gold do not react with water at all.
ii. Reaction of non-metals with water:
Most of the non-metals do not react with water, but most of the non-metals are reactive in air.
Non-metal + Water \(\to\) No reaction
Reaction of materials with air:
Air is an invisible artist; it holds secret powers. Metals and non-metals react differently with air, revealing the secret of oxygen, which is quietly influencing the materials around us.
Let us learn the reactions of metals and non-metals with air. Observing these reactions helps us understand the unique properties of different materials.
Activity: Burning of magnesium ribbon
Materials required:
- Magnesium ribbon (\(2\) - \(3\) cm long)
- Two tongs
- Burner
- Two goggles
- Watch-glass
- Red and blue litmus papers
- Distilled water
- Beaker and sandpaper
Experimental procedure:
Step 1: Take a magnesium ribbon (2 - 3 cm long) and clean it with sandpaper. This cleaning will remove the oxide layer on the magnesium ribbon, making it active.
Step 2: Hold the magnesium ribbon over a watch glass with tongs and burn it in the air using a burner (as shown in the figure). Wear a pair of dark-coloured goggles, watch the burning magnesium ribbon.
Step 3: Take a watch glass and collect the white ashes (magnesium oxide).


Burning of magnesium ribbon
Observation:
- We observe that the magnesium ribbon burns with a dazzling white flame.
- The magnesium ribbon is converted into a white powder (collected in the watch glass).
Result:
The white powder collected after burning the magnesium ribbon is magnesium oxide (\(MgO\)).
Litmus test:
- Transfer the white powder to a beaker with a small amount of distilled water, then mix it thoroughly.
- Pour a few drops of the mixture into the watch glass, and place the blue and red litmus paper on it.


Litmus test of magnesium oxide
Among the red and blue litmus papers, the colour of red litmus paper changes to blue. The colour of blue litmus paper remains the same (no change).
The change in the colour of red litmus paper to blue suggests that the aqueous solution of magnesium oxide is basic in nature. Most metals react with oxygen to form metal oxides. Most metal oxides are basic in nature.
Metal + Oxygen \(\to\) Metal oxide
However, the reactivity of metals towards oxygen differs. This depends on the reactivity of the metals,
- Sodium and potassium react vigorously with oxygen.
- Magnesium and calcium burn slightly above room temperature.


Reactivity series
- Iron does not burn when heated, but iron filings do when put into the burner's flame. Copper does not burn, but it does leave a black film of copper (II) oxide on the hot metal.
- Gold and platinum do not combine with oxygen even at high temperatures.
Metals like potassium and sodium react rapidly with oxygen and readily catch fire in the air. As a result, they are immersed in kerosene oil to protect them and prevent accidental fires.
Effect of air and water on non-metals
Activity: To find the chemical properties of non-metals
- Take a small amount of sulphur powder on a spatula, and heat it over a burner.
- To collect the liberated gas, place an inverted test tube over the spatula.
- Carefully remove the test tube without allowing the gas to escape.


Burning sulphur powder on a spatula
Reaction:
Heating sulphur produces sulphur dioxide gas.
Litmus test:
Fill the test tube with distilled water and shake it. On a watch glass, add a few drops of the solution and place blue and red litmus paper on it.


Litmus test of sulphur dioxide
Among the red and blue litmus papers, the colour of the blue litmus paper changes to red. The colour of red litmus paper remains the same (no change).
The change in the colour of blue litmus paper to red suggests that sulphur dioxide is acidic in nature.
Non-metals react with oxygen to form non-metal oxides. Non-metal oxides are mostly acidic in nature.
Non-metal + Oxygen \(\to\) Non-metal oxide
Some non-metals such as sulphur and phosphorus react rapidly with oxygen and readily catch fire in the air. As a result, they are stored in water to protect them and prevent accidental fires.
Reference:
https://th.bing.com/th/id/OIP.2q8wHQBtT6cTiWp7Lx6F6AAAAA?pid=ImgDet&rs=1
https://live.staticflickr.com/6091/6257265455_aed62037cc_b.jpg