Everything in this universe that has space and mass is made of matter. The study of the nature of matter explores how these tiny, invisible particles combine to form the things around us. It reveals the secret behind our world and ourselves.
Nature of matter:
Matter is made up of large number of particles. The particles of matter are very tiny and are beyond our imagination. The particles of matter have mass and occupy space. Matter is made up of atoms, which combine to form molecules and substances.
An atom is the smallest particle that cannot be seen with the naked eye, even with standard microscopes. Modern instruments such as the SEM (Scanning Electron Microscope) and TEM (Transmission Electron Microscope), use beams of electrons to study very small structures.
Activity: Breaking chalk into smaller particles
Step 1: Take a piece of chalk and break it into two halves.
Step 2: Continue breaking the chalk into smaller and smaller pieces until it becomes hard to break with your hand.
Step 3: Now, collect the smaller pieces and grind them using a mortar and pestle to obtain a fine powder.
Step 4: Observe the fine chalk powder using a magnifying glass.

Chalk piece and ground chalk powder
The specks of chalk powder can be ground further into even tinier particles. The grinding process continues can make the particles smaller, but they cannot be broken down indefinitely by physical methods.
These tiny units of chalk are the basic building blocks of a whole piece of chalk, which is composed of a large number of smaller units. These smaller units are called the constituent particles. Each tiny particle still retains the properties of the chalk.
A constituent particle is the basic unit (smallest unit) that makes up a larger piece of a substance or a material. All matter around us is made up of extremely tiny particles called constituent particles. Though the constituent particles are very small, they retain the properties of the substance they combine to form.
Activity: Dissolving sugar in water
- Take a clean glass tumbler and fill it with drinking water.
- Add two spoons of sugar into the water without stirring.
- Taste a small amount of water from the top layer and observe its taste.
- Now stir the water until the sugar dissolves completely, then taste the water from the top layer and observe its taste.
Observation:
Before stirring, the top layer of water does not taste sweet. After stirring, the top layer of water tastes sweet.
Conclusion:
Sugar dissolves completely in water and seems to disappear. But the water tastes sweet, proving that the constituent particles of sugar spread uniformly throughout and occupy the available spaces between the particles of water without losing their properties. These spaces between the particles are known as interparticle spaces.
Characteristics of particles of matter:
The characteristics of particles of matter are as follows:
i. Particles of matter have space between them.
ii. Particles in matter are continuously moving.
iii. Particles of matter attract each other.
Take a beaker of water and add some salt. Stir the mixture using a glass rod until the salt dissolves completely. Observe the level of water in the beaker. it is not raised.

Dissolving salt in water
The particles of matter are very tiny in nature. When salt dissolves in water, its particles occupy the spaces between water. This proves that there is space between the particles of matter. Similarly, when making lemonade, tea or coffee, the same principle applies.
Particles in the matter are continuously moving:
The particles of matter are always in continuous motion. To understand this concept, let us perform a simple experiment.
The movement of particles is dependent on temperature. The kinetic energy of the particles increases with increasing temperature.
i. When the temperature increases, particles gain more energy and move faster.
ii. When the temperature decreases, particles lose energy and move slowly.
i. When the temperature increases, particles gain more energy and move faster.
ii. When the temperature decreases, particles lose energy and move slowly.
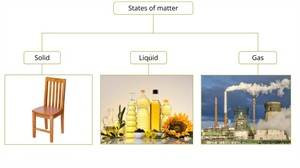
States of matter
A building or house is constructed with the help of bricks, stones and other solid materials. And, the water we drink, the lake, and the sea are in liquid form. Then, the air we breathe and the gases in our atmosphere are in a gaseous state.
Therefore, generally, we can classify the matter into three types:
Therefore, generally, we can classify the matter into three types:
- Solid state
- Liquid state
- Gaseous state
States of matter
Solid state:
Objects that which have a definite shape, distinct boundaries, and a fixed volume are called solid objects.
Example:
A book, pen, pencil.
A car, bike, plane, bus, etc.
The solid objects tend to retain their original shape despite external forces.

Rocks
- Solids have definite shape and volume.
- The particles of solids are very closely packed, and the interparticle attractions are very strong.
- The strong force of attraction holds the particles in a fixed position, preventing them from moving freely.
- The particles do not have enough space to move from one place to another. Instead, they can only vibrate or oscillate about their fixed positions.
- The molecules in the solids are so closely packed that they are rigid and maintain a definite shape.
For example, if we apply external force to the piece of chalk, it will break into pieces, but the nature of the particles will not be altered, as solids are rigid bodies.
Intermolecular force on solids:
The intermolecular force in the solid is the strongest force compared to the liquid and gas. This strongest intermolecule force makes solid matter denser and gives it a certain shape and structure.
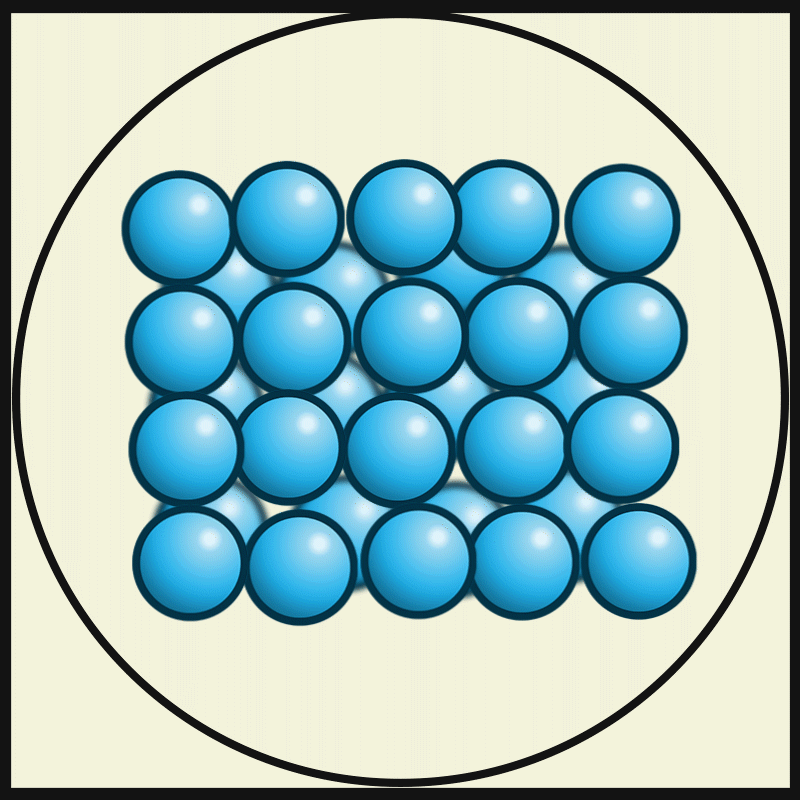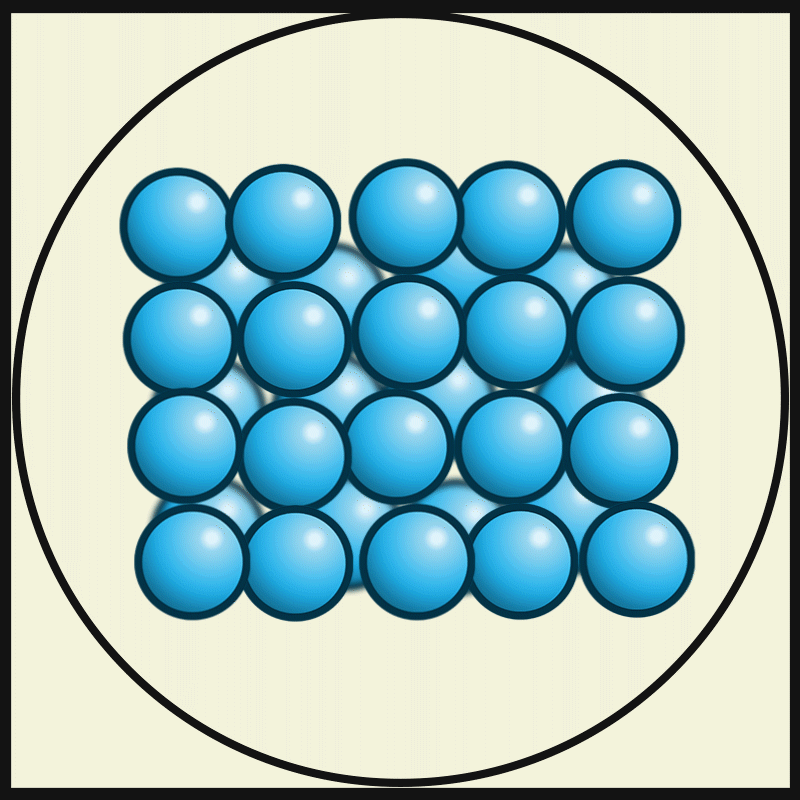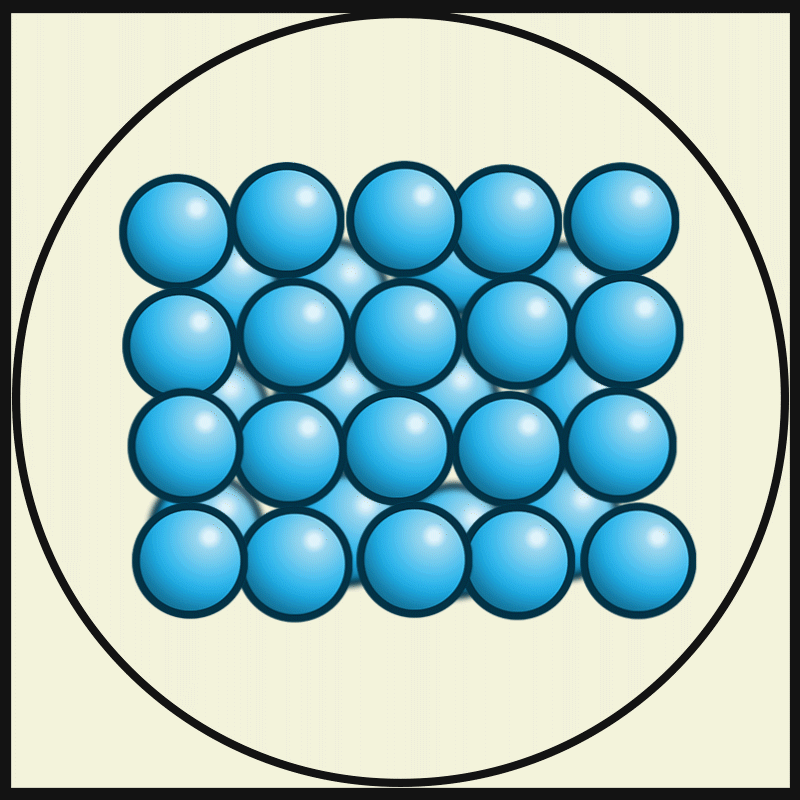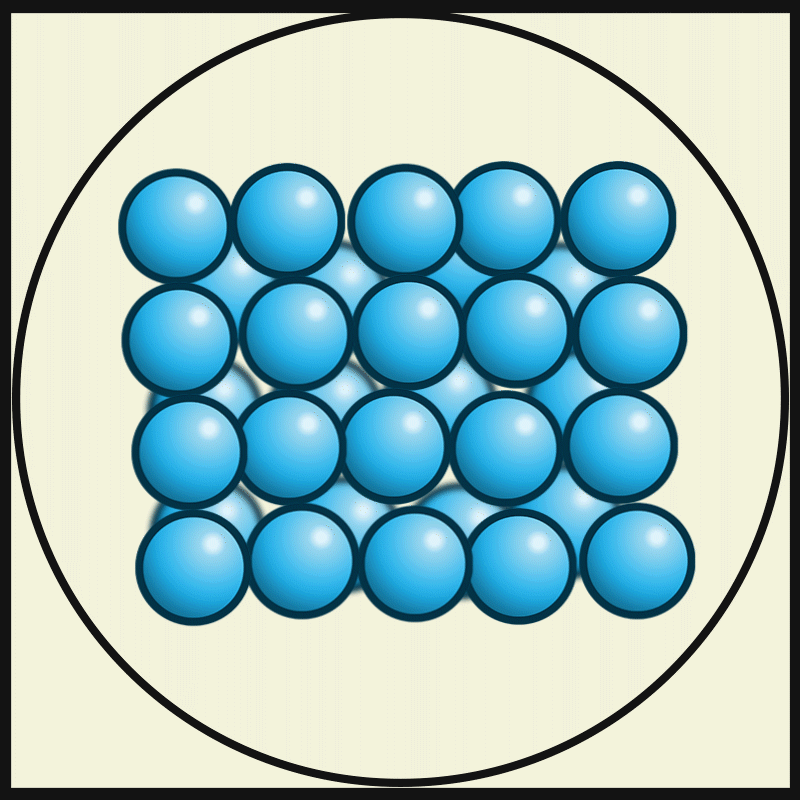
Interparticle spacing in solids
Effect of temperature on solids:
When a solid is heated, its particles absorb energy and their kinetic energy increases, and they begin to viberate vigorously. As the temperature increases, the vibrations become so vigorous, that the particles overcome the interparticle forces of attraction, and the solid converts to a liquid state.
The minimum temperature at which a solid melts into a liquid under atmospheric pressure is called the melting point.

Melting of ice cubes
The melting point of ice is .
Some solids have weak interparticle forces of attraction, so their melting points are low. At the same time, others have strong attractive forces and have high melting points.
Example:
i. Urea - \(133°C\)
ii. Iron - \(1538°C\)
Liquid state:
In our day-to-day life, we come across many liquid states, such as the water we drink, the oil we use for cooking, and the fruit juice we often drink. These are all the matters in a liquid state.

Fruit juices
Liquids do not have a specific shape, but they can take on the shape of the container when we pour them in the container. For example, we might have seen the juice bottles. The liquid inside the bottle takes the shape of the bottle.
- The liquid is an intermediate phase between the solid and gas.
- Liquids have a fixed volume but no fixed shape.
- Liquids take the shape of the container without changing their volume.
Intermolecular Force on Liquid:
Activity:
Take some water and try moving your fingers through it. Fingers can move through water without breaking or cutting it permanently: the same cannot be done with solids.

Moving fingers through water
As soon as the finger is removed, the water return to its original position.
The interparticle force in a liquid is not as high as that in a solid, but it is still strong enough to keep the particles closer.
Interparticle space of states of matter
Effect of temperature on liquids:
When a liquid is heated, the kinetic energy of the particles increases, and they move faster. As the temperature increases, the force of attraction between liquid particles decreases, allowing them to move more freely. Eventually, the constituent particles gain energy, and the liquid is converted into vapour or the gaseous state.
The temperature at which a liquid begins to boil and turns into vapour at the atmospheric pressure is known as its boiling point.
Gaseous state:
While your mother cooks in the kitchen, you can sense the delicious aroma coming to you even though you're in another room. Have you ever wondered how that smell came to you?
And, we all like balloons, right? But, have you ever wondered how the balloon seller fills so many balloons in a short span of time? The answer is gases.

Gas characteristics
Gases are highly compressible as compared to solids and liquids. In the gaseous state, the particles move about randomly at high speed.
Because of this property, we can use LPG in a highly compressed condition. If we compress the gas particles, we can store them in a small container too.
We know that gas particles are more freely movable, so that they can travel through the air more easily. That's why the aroma of a food reaches you even though you're in a separate room.
Intermolecular force on gases:
The intermolecule force in the gaseous substance is weaker compared to that in liquid and solid. This weaker intermolecular force makes the gaseous matter to move freely in the air.
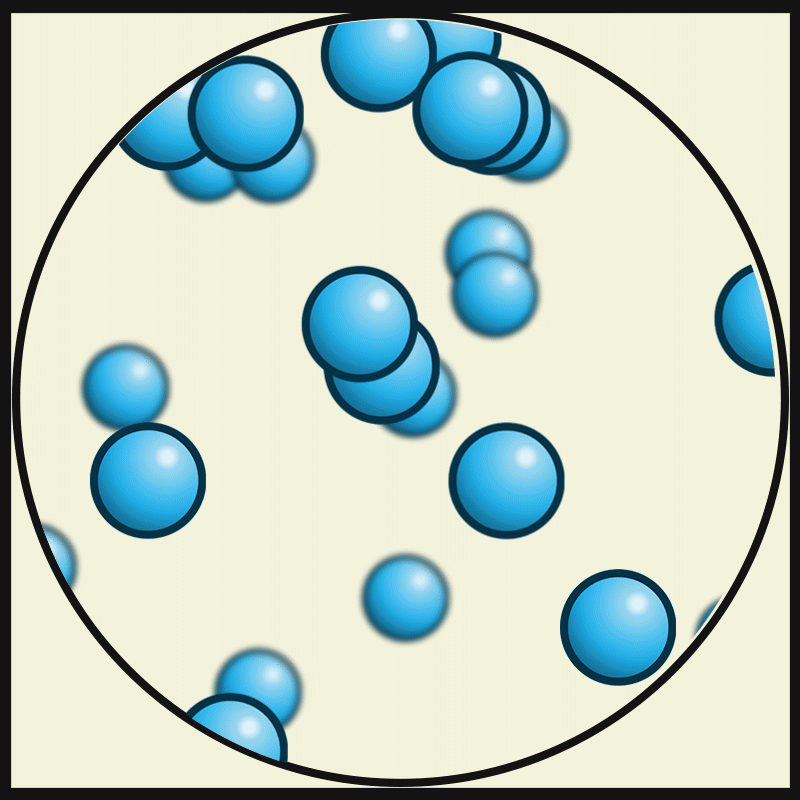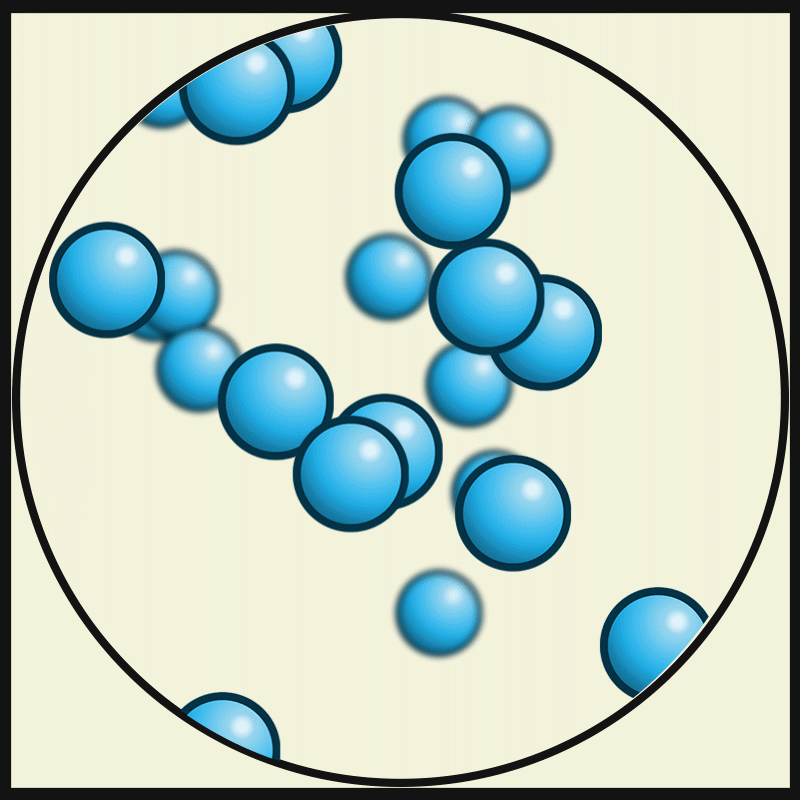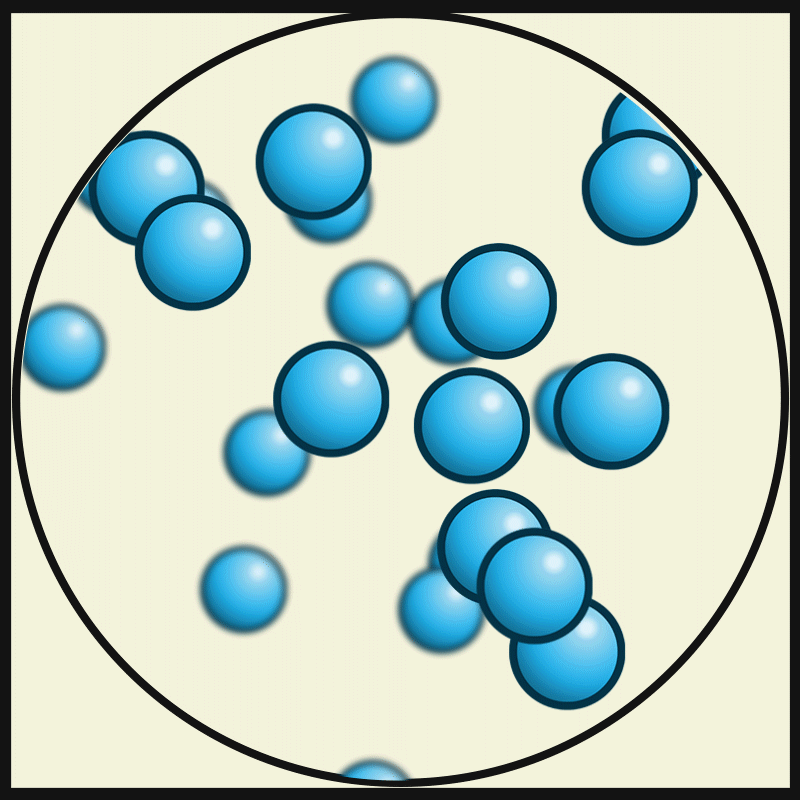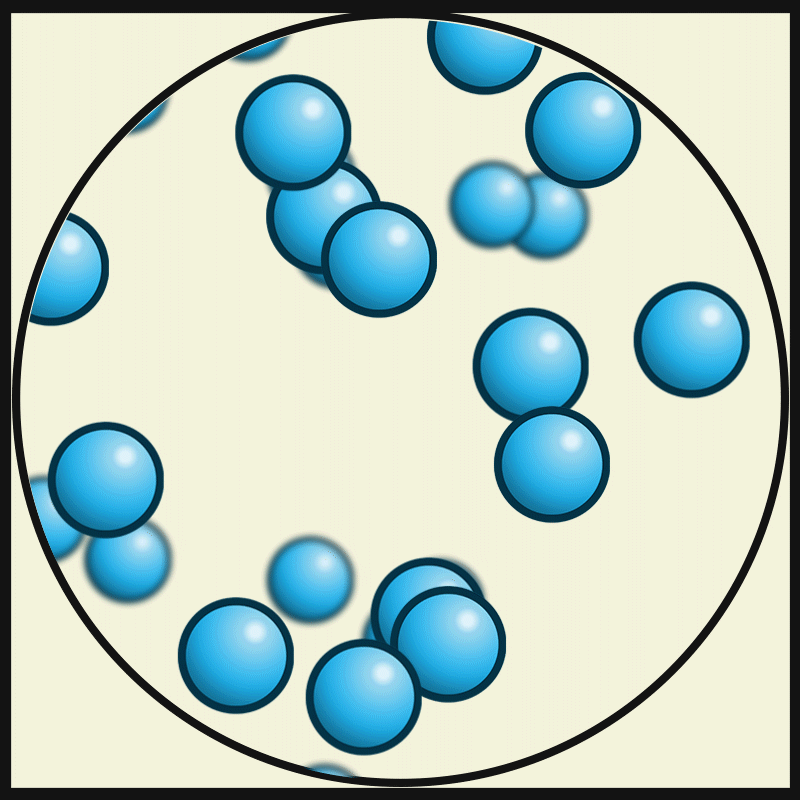
Interparticle spacing in gases
Transformation of states:
Have you ever wondered why ice turns into puddles of water on a sweltering day?
Knowing that stated can change, when a state change occurs, the properties of substances also change. Similarly, if the state change is reversed, the substance will recover the properties which it had before.

Transformation of states of matter
Matter changes its state with respect to the energy applied to it. The following figure shows how matter changes state wherever the energy is applied.
Transformations of states
This leads to a rearrangement of the substance because the attractive force no longer holds the particles tightly.
Reference:
https://commons.wikimedia.org/wiki/File:VW_Fingers_touching_water.jpg
By Julio Miguel A Enriquez and Monica Muñoz - Wiki Learing Tec de Monterrey, CC BY-SA 4.0, Link