Compressibility of states of matter:
Compressibility is the measure of the ability of a substance to decrease the volume of a substance when pressure is applied to it.
Gases are highly compressible compared to solids and liquids due to the larger space between the particles (or ions or molecules) in them.
Compressibility of states of matter
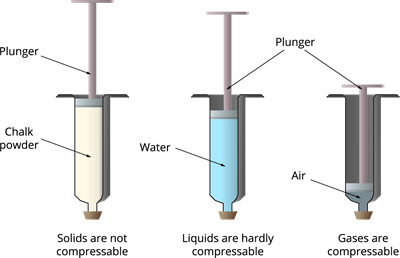
Consider three syringes filled with chalk powder, a liquid and a gas. On pressing the plunger down, the pressure is applied, and the following changes will occur:
i. A syringe with chalk powder: Cannot be compressed easily since the solid particles are packed tightly.
ii. A syringe with liquid: Hardly compressible because the particles are already close together.
iii. A syringe with gas: Highly compressible since they have a large amount of empty spaces.
i. A syringe with chalk powder: Cannot be compressed easily since the solid particles are packed tightly.
ii. A syringe with liquid: Hardly compressible because the particles are already close together.
iii. A syringe with gas: Highly compressible since they have a large amount of empty spaces.
Liquefaction:
The process of converting a gaseous state substance into the liquid state by applying pressure and lowering its temperature is known as liquefaction.
When pressure is applied, and temperature is lowered, gas molecules come closer together. Lowering the temperature removes energy from the molecules, helping the gas change into a liquid.
Interparticle spacing in states of matter:
Take a beaker with water and dissolve some salt with the help of a glass rod. Wait for a few seconds.
We already know that particles of matter are very tiny. When we dissolve salt in water, the particles enter the spaces between the water particles. Thus, the water level does not raise significantly.

Dissolving salt in water
When we dissolve salt in water, the particles of salt occupy the spaces between the particles of water. This experiment clearly shows that there is enough space between the particles of matter.
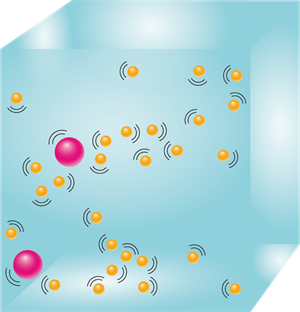
Distribution of salt particles in water
And think about what happens when adding insoluble solids, such as sand or stone, instead of salt or sugar. If we imagine adding sand, the water level will rise.

Sand in water
Sand is an insoluble solid; it does not dissolve in water. The sand particles settle at the bottom and occupy space in the container, causing the water level to rise.
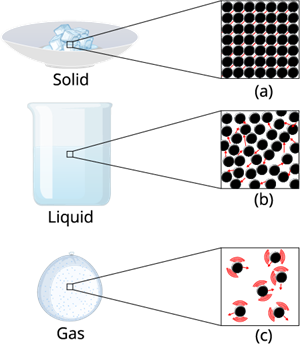
i. The constituent particles of solids are held together by a strong force of attraction. As a result, the particles cannot move freely from one place to another. Though the particles of a solid are closely packed, there is still a tiny space present between them.
Interparticle spacing in states of matter
ii. In liquids, the forces of attraction between particles are moderate and weaker than in solids. As a result, the particles are not fixed in one place and can move past one another. This allows liquids to flow.
iii. In gases, the forces of attraction between particles are very weak. The particles are far apart,with very large interparticle spacing. These large spaces allow gas particles to move freely in all directions.
Diffusion and movement of particles of states of matter:
The movement of particles is the key reason for the differences in the properties of solids, liquids, and gases.
Diffusion is the process by which particles of a substance move from a region of higher concentration to a region of lower concentration due to their constant random motion, until they are evenly spread.
Example: A drop of ink spreads in water even without stirring.

Diffusion of particles in water
Activity: To demonstrate the diffusion of a solid in water
- Take a glass beaker filled with clean water.
- Add a few crystals of copper sulphate to the water.
- Do not stir the water and observe the changes.

Diffusion of copper sulphate in water
Observation:
The water near the crystals turns blue first. Gradually, the blue colour spread throughout the entire water.
Copper sulphate crystals spread in water due to diffusion. Diffusion occurs faster in liquids because their particles have space between them and are in constant motion.
The diffusion of particles increases with increasing temperature. Also, when the temperature is increased. The kinetic energy of the particles increases with increasing temperature.
- When the temperature increases, particles gain more energy, move more quickly, and dissolve faster.
- When the temperature decreases, particles lose energy, move more slowly, and dissolve slowly.
Movement of gas particles:
Burn an incense stick in one corner of the room; initially, the smell is felt only near the incense stick.

Burning of an incense stick
After a while, the fragrance spreads and becomes detectable throughout the room. This happens because the fragrance particles spread, filling the entire room. This process shows that particles in gases are continuously moving and diffusing.
Similarly way, the smell of a perfume sprayed in one part of a room spreads to other parts after some time.
The aroma of hot food spreads faster in a room than that of cold food because particles move faster at higher temperatures.