Factors affecting the solubility:
- Nature of the solute and solvent
- Temperature
- Pressure
1. Nature of the solute and solvent:
The nature of the solute and solvent greatly influences solubility. Although water dissolves a wide range of ionic and covalent substances, it does not dissolve all subsrances. A solution in which water acts as the solvent is called an aqueous solution.
To predicting solubility, scientists commonly use the phrase "like dissolves like". This means that a substance dissolves best in a solvent that has a similar nature. Polar solutes dissolve in polar solvents, while non-polar solutes dissolve in non-polar solvents.
For instance, common salt is a polar compound that dissolves easily in a polar solvent such as water, but it does not dissolve in non-polar solvents such as oil.
Similarly, non-polar solutes dissolve in non-polar solvents. For example, fat dissolves in ether.

Iodine in a) carbon tetrachloride b) water
Iodine is a non-polar substance; so it dissolves in non-polar solvents such as carbon tetrachloride. However, it does not dissolve in the polar solvent water. Thus, Non-polar compounds do not dissolve in polar solvents, and polar compounds do not dissolve in non-polar solvents. This principle is explained by the rule “like dissolves like.”
2. Temperature:
Solubility of solids in liquid:
In general, the solubility of a solid solute in a liquid solvent increases with an increase in temperature.
Dissolution is the process in which a solid solute dissolves completely in a solvent to form a homogeneous solution.
The solubility of the solute generally increases with an increase in temperature. When the temperature is increased, a saturated solution maybecome unsaturated solution, allowing more solute to dissolve.
The kinetic energy of the particles increases with increasing temperature.
- As the temperature increases, particles gain more energy, move faster, and the solute dissolve more quickly in the solvent.
- As the temperature decreases, particles lose energy and move more slowly. and ther soluter dissolves more slowly.
Example: Sugar dissolves more readily in warm water than in cold water.
Based on the change in temperature during the process,
- In an endothermic process, solubility increases because heat is absorbed.
-
In an exothermic process, solubility decreases because heat is released.
Solubility of gases:
Many gases are dissolved in water. Oxygen dissolves in water only in small quantities, even this smaller quatity is very important. The dissolved oxygen in water helps sustain aquatic life such as plants, fish, and other organisms.
The solubility of gases is the ability of a gas to dissolve in a liquid to form a solution under specific conditions of temperature and pressure.


Aquatic species in water
When gases dissolve in water, they form a homogeneous (uniform) mixture. Gases such as oxygen and carbon dioxide dissolve in water and are evenly distributed throughout the liquid, resulting in a solution.
In contrast, if gas bubbles are visible and not completely dissolved, the mixture is heterogeneous (non-uniform). As long as the gas remains completely dissolved in the liquid, the mixture is a homogeneous (uniform) solution. Among common gases, ammonia is the most soluble in water.
Factors affecting the solubility of gases in liquids:
i. Temperature:
In general, water contains dissolved oxygen. When water is heated, the solubility of oxygen decreases, allowing it to escape as bubbles.
Aquatic animals thrive in colder climates because the water contains a higher concentration of dissolved oxygen. This demonstrates that oxygen is more soluble in water at low temperatures.
ii. Pressure:
Only when gas is soluble in a liquid are pressure effects observed. The solubility of a gas in a liquid increases with increasing pressure.
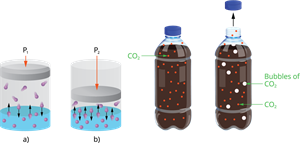

Effect of pressure on solubility
Example: Carbonated beverages, i.e. soft drinks, household cleaners containing an aqueous solution of ammonia, formalin -aqueous solution of formaldehyde, and so on, are common examples of gas solubility in liquids.
Density of materials:
In a beaker filled with water, an iron ball and a wooden cork are dropped simultaneously. Now, what will you observe? From the picture, it is observed that the cork floats and the iron ball sinks.


Floating and sinking of objects
When a single drop of water is dropped into the oil, it sinks. However, if one drop of oil is added to water, it floats to the surface. As a result, some oils are denser than water.


Oil floating on water
Water is denser than cooking oil and castor oil, even though these oils appear to be denser than water. Castor oil has a density of \(961 kg/m^3\), whereas water has a density of \(1000/ kg/m^3\) or \(1/ g/cm^3\).
Density is defined as the amount of mass present in a unit volume of a substance.
Materials with a higher density are denser, while those with a lower density are less dense or lighter. The density of a substance is independent of its shape or size.
The density of a substance may be expressed mathematically using the formula:
The SI unit of density is \(kg/ m^3\) and the CGS (Centimetre Gram Second) unit is \(g/ cm^3\). The common unit of density for liquids is expressed as \(g/mL\).
Reference:
https://commons.wikimedia.org/wiki/File:CNX_Chem_13_01_SuperSat.jpg
https://media.easy-peasy.ai/27feb2bb-aeb4-4a83-9fb6-8f3f2a15885e/77fdae37-0aec-40fa-9a0d-dbed63d52f1b.png