Have you ever wondered why salt seems to disappear when added to water and stirred well? The salt crystals vanish, yet the salty taste remains. The reason is that salt dissolves in water, forming a solution. The salt particles breaks down into individual ions (\(Na^{+}\) and \(Cl^{-}\)) and spread uniformly throughout the water, making the solution homogeneous.
The world is full of solutions: the coffee we drink, the blood in our veins, the air around us, the lemonade, and even medicines. All these are examples of solutions. The study of solutions tells the story of molecular friendship and cooperation between particles.

Solutions
Let us learn how substances interact, combine, and dissolve to form a solution and understand their nature.
Types of mixture:
The components of the mixture generally do not undergo any chemical reactions with one another. In some mixtures, the components can be seen, while in others, they are too small to be seen with the naked eye.
Based on the visibility and nature of their components, mixtures are classified into:
- Uniform mixture
- Non-uniform mixture
1. Uniform mixture:
- All components of the mixture blend evenly.
- There are no visible separate barriers; the mixture appears uniform throughout.
- It is a single-phase mixture.
- Example: Salt completely dissolved in water, sugar completely dissolved in water
- Uniform mixtures are also called homogeneous mixtures.

Uniform mixture
2. Non-uniform mixture:
- All components of the mixture are unevenly distributed.
- There are noticeable separate barriers between components.
- There are two or more phases in the mixture.
- Example: Oil and water, sand and water, and salad
- Non-uniform mixtures are also called heterogeneous mixtures.
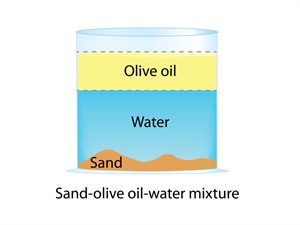
Non-uniform mixture
Solute:
A solute is the substance that dissolves in the solvent to form a solution, which is a homogeneous mixture. It is usually present in a smaller quantity, and its key characteristic is that it dissolves.

Solute
A Solute can exist in different states of matter:
- Solid solute: Salt, sugar
- Liquid solute: Acetic acid
- Gas solute: Carbon dioxide, oxygen
Solvent:
A solvent is a substance that dissolves the solute particles to form a solution. The solvent is usually present in a larger quantity than the solute.
Water is called a universal solvent because it can dissolve many substances. It is a polar solvent, and solutions in which water is the solvent are called aqueous solutions.

Universal solvent: Water
Some substances do not dissolve in water. To dissolve such substance, non-polar solvents such as ethers and benzene are used.
Solution:
Whenever we drink lemonade juice, it tastes the same throughout. This shows that particles of sugar (or salt) are evenly distributed in the solution (juice).
A solution is a homogeneous mixture of two or more substances that appears to be uniform and has the same or similar properties throughout.
When a solute is dissolved in a solvent, a solution is formed.
\(Solution = Solvent + Solute\)

Formation of a solution
A solution must have at least two components: a solute and a solvent. Binary solutions are those that consist of one solute and one solvent (two components).
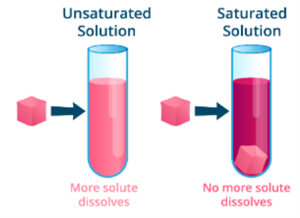
Saturated solution:
A saturated solution is one in which no more solute can be dissolved in a specific amount of solvent at a given temperature.
Example:
At \(25° C\), \(36 g\) of sodium chloride can dissolve in \(100 g\) of water to form a saturated solution. If some more sodium chloride is added beyond this, it will remain undissolved because the solution has reached its solubility limit.
Saturated and unsaturated solution
Unsaturated solution:
An unsaturated solution is one in which the solvent can still dissolve more solute at a given temperature.
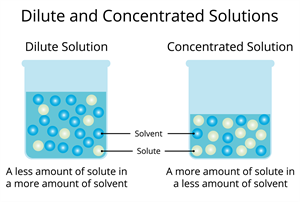
Concentrated solution:
A concentrated solution contains a large amount of solute dissolved in a given amount of solvent. In such solutions, the quantity of the dissolved solute is much higher compared to the solvent.
Dilute solution:
A dilute solution contains a small amount of solute dissolved in a given amount of solvent. In this case, the quantity of the dissolved solute is much lower compared to the solvent.
Types of solution based on solute
When two solutions contain the same solute and solvent, the solution with more solute is concentrated, and the one with less solute is dilute.
The difference between dilute and concentrated solutions can be seen by colour, density, or taste.
Comparison of dilute and concentrated solutions:
The dark-coloured solution of tea or copper sulphate is the concentrated solution, while the lighter-coloured solution is the dilute solution.

Concentrated solutions

Dilute solutions