Liquids do not have a definite shape. But they take the form or shape of the container they are stored in. So, even though liquids have definite volume, it is not easily determined, unlike solids.

Liquid adapts the shape of the container
Volume is the measure of the amount of space occupied by an object. The SI unit of volume is \(cubic\ metre\) (\(m^3\)).
Volume is the measurement of three-dimensional space occupied by an object. Volume of smaller objects is expressed as \(cubic\ decimetre\) (\(dm^3\)) or \(cubic\ centimetre\) (\(cc\)) (\(cm^3\)).
The volume of an object can be determined from the density formula by dividing the mass of an object by its density.
Unit to measure the volume of liquid:
A \(litre\) (\(L\)) is a unit of measurement used to express the volume of liquids. For example, \(2\ litres\) oil packet, \(20\ litres\) water bottle and so on. Other units are \(millilitre\) (\(mL\)), \(centilitre\) (\(cL\)), and \(kilolitre\) (\(kL\)).
The readings in the measuring beakers are usually marked in units of a \(millilitre\) (\(mL\)). In terms of a \(millilitre\),
- \(1\ L\) \(=\) \(1000\ mL\)
- \(1\ mL\) \(=\) \(0.001\ L\)
- \(1 mL\) \(=\) \(1\ cm^3\)
Liquids can be measured in \(litres\) and can also be expressed in \(cubic\) \(metre\) (\(m^3\)).
Measuring tools:
A liquid can be poured into a graduated container to easily determine its exact volume. There are various measuring tools to find the volume of a liquid. Some of them are,

Measuring cylinders
A measuring cylinder is one of the common apparatus used to measure the volume of liquids. It is a narrow, cylindrical and transparent container. It has graduated markings to indicate the volume of liquid.
Measuring cylinders are available in different capacities, such as \(10\ mL\), \(20\ mL\). \(50\ mL\), \(100\ mL\), \(200\ mL\), \(250\ mL\), and \(500\ mL\)
The volume of a liquid is the amount of space it occupies in a container. Therefore, the readings marked on the measuring containers directly indicate the volume of liquid.
Activity: To determine the volume of liquid in a measuring cylinder
Take a measuring cylinder and observe it.

Measuring cylinder 100 mL
Observation of a measuring cylinder:
i. Maximum volume of the cylinder: The cylinder is marked up to \(100\ mL\), hence its maximum capacity is \(100\ mL\).
ii. The volume difference between the two bigger marks: The two successive big marks have a volume difference of \(10\ mL\).
Example: From \(10\ mL\) to \(20\ mL\)
iii. Smaller divisions: The number of smaller divisions between the two successive bigger marks is \(10\)
iv. Least count: One small division can read the volume of \(1 mL\)
The smallest volume that can be represented by the \(100\ mL\) measuring cylinder is \(1 mL\).
Therefore, the least count is \(1\ small\ division = 1\ mL\)
Similarly, the least count varies depending on the capacity of the measuring cylinder.
- Measuring cylinders such as \(10 mL\) or \(25 mL\) can measure as small as \(0.1 mL\).
- The least count of a \(250 mL\) capacity cylinder is \(2 mL\).
- The least count of a \(500 mL\) capacity cylinder is \(5 mL\).
Thus, smaller-capacity measuring cylinders give more accurate values.
Measuring liquid using a measuring cylinder:
Let us measure \(20\ mL\) of water,
- Take a clean, dry measuring cylinder and place it on a flat surface.
- Pour water into the measuring cylinder until the required level is reached.
- If the required water level is above or below the mark, adjust it by adding or removing a few drops of water with a dropper.
- On careful observation, you will notice that the surface of water is curved. This curved surface is called a meniscus.

Meniscus at 20 ml
- For colourless liquids, take the reading from the bottom of the meniscus.
- While taking the reading, keep your eye at the same level as the bottom of the meniscus to minimise error.
- Once the water level reaches the required volume of \(20\ mL\), transfer it to the required container.
In case of coloured liquids, the readings should be taken from the top of the meniscus. Example: Mercury
Volume of solid objects with regular shapes:
Collect various objects in a cuboid shape to determine their volume. The objects, such as notebooks, boxes, bricks, and dice. Using a measuring scale, measure the lenght (\(l\)), breadth (\(b\)), and height (\(h\)) of the objects.
Calculate the volume of cuboid objects using the formula,
Volume of objects with irregular shapes:
There is no exact formula for measuring irregularly shaped objects, but there are methods that can be used. Since volume is the total space occupied by an object, the volume of a small irregular piece of stone can be found by the water displacement method.
Activity: Water displacement method
The volume is determined by using a measuring cylinder, a piece of stone, a thread and water.

Water displacement method
Procedure:
- A measuring cylinder with markings is filled with water.
- From the readings of the measuring cylinder, the volume is taken as \(V_{1}\).
- A stone is tied to a piece of thread and fully immersed in water.
- The thread is held in such a way that it does not touch the cylinder walls.
- When the stone is immersed, the water level rises.
- Again, the readings of the measuring cylinder are noted, and the volume is taken as \(V_{2}\).
Inference:
The stone occupied space inside the cylinder by displacing some water. This made the water level rise. Therefore, the volume of water displaced will be equal to the volume of space occupied by the stone in the container. This technique is known as 'the water displacement method' and was discovered by Archimedes.
Formula:
\(V_1\) - Initial reading
\(V_2\) - Final reading
Layers of Earth:
The Earth is composed of several layers, including the crust, the upper mantle, the lower mantle, the outer core, and the inner core. Each layer has a specific density range.
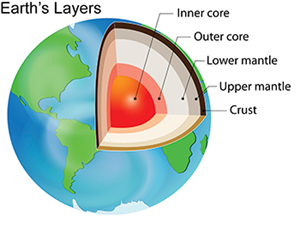
Layers of Earth
The outer layer of Earth is called the crust, and it is the lightest layer. The density increases as we move deeper towards the centre. The inner layers are heavier. On moving deeper into the Earth, both temperature and pressure increase slightly, making the materials denser and more tightly packed.
Effect of temperature on density:
i. Heating:
On increasing the temperature, the density of the substance decreases. When a substance is heated, its particles gain energy and move faster. The particles tend to move far away, increasing the volume. As volume increases and mass remains constant, the density of the substance decreases.
ii. Cooling:
On decreasing the temperature, the density of the substance increases. When a substance is cooled, its particles cool and move closer together. As the particles tend to move closer, the volume decreases. As the volume decreases and mass remains constant, the density of the substance increases.
Heating: Volume increases, mass remains constant, and density decreases.
Cooling: Volume decreases, mass remains constant, and density increases.
The hot air balloon works on the same principle. When the burner is turned on, the air inside the balloon becomes less dense than the air outside. Because the air around the hot air balloon is denser, it pushes the balloon upwards.

Hot air balloons
The hot air is lighter than the cold air. When the burner is turned off, the air inside the balloon cools, it becomes denser, and the balloon comes down.
Effect of pressure on density:
The effect of pressure on density depends on the different states of matter.
i. Gases: Particles of gases are loosely packed and far apart. As pressure increases, the particles move closer together, decreasing the volume and increasing the density.
ii. Liquids: Particles of liquids are moderately packed, almost close together, and they are almost incompressible. Thus, the change in density is very small.
iii. Solids: Particles of solids are tightly packed with strong intermolecular forces, and they are incompressible. Thus, the change in density is negligible.
Reference:
https://commons.wikimedia.org/wiki/File:Hot_air_balloons_in_leon.jpg
https://freesvg.org/graphic-showing-the-meniscus-vector-image